|
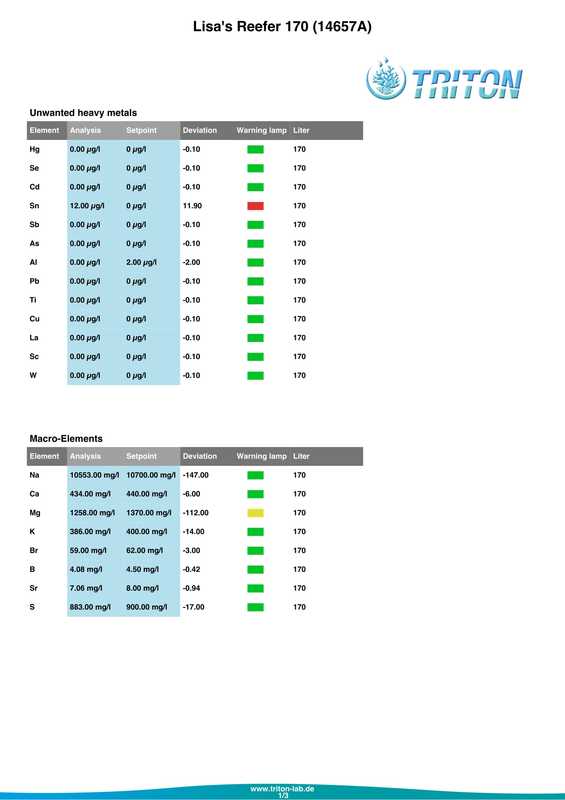
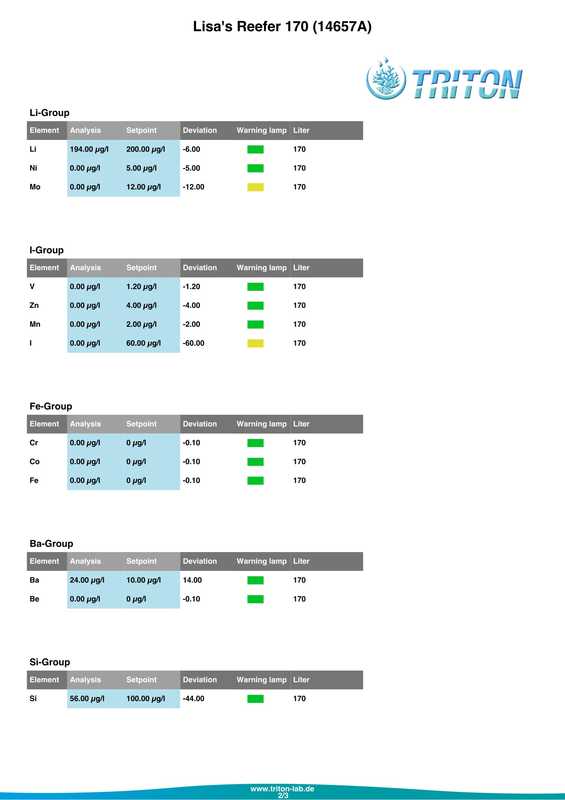
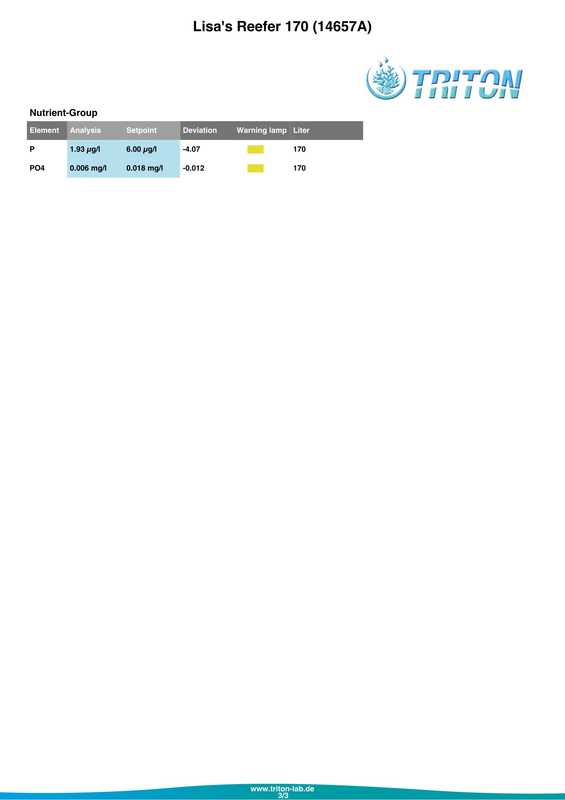
In my quest to discover why some of my corals were looking pale (in one case very pale indeed) with poor or non existent polyp extension I decided to send a sample of tank water off to Triton for an ICP-OES (which stands for Inductively Coupled Plasma-Atomic Emission Spectroscopy) analysis. I must admit that I had actually intended to do this anyway when the tank reached the 6 month mark just because I was curious as to what it would tell me about my water quality. At the same time as sending off the sample I did a whole round of testing myself. The intention being that I could compare both results and see if my test kits (not to mention my testing technique) were up to scratch. Here are the results from my testing performed on the 18th April 2017. Specific gravity: 1.026 (51.7mS according to my conductivity probe) Alkalinity: 7.0dKH (Salifert) Calcium: 410ppm (Salifert) Magnesium: 1272ppm (Salifert) Potassium: 390ppm (Salifert) Strontium: 0-3 (Salifert) Nitrate: undetecable (Salifert) Phosphate: undetectable (Hanna phosphate) Iodide: <0.01 (Salifert) Iodate: <0.03 (Salifert) ...and here are the ICO-OES results. So what do the above results tell me? Well, the obvious issue that immediately jumps out is the elevated tin (Sn) level. Eeek! Where on earth did that come from? I have read that a possible source for tin could be the glass (as molten tin is used to "float" the glass on during it's manufacture). Other than that, something could be rusty and contaminating the system but I'm at a loss as to what it could be, all the equipment was bought new and there's nothing obviously rusting as far as I can see.
Apart from tin, I don't think the rest of the results look too bad really, pretty much as I expected. My tests are not that far off which is good to know (with the possible exception of Strontium which is low with the Salifert test kit but actually at an acceptable level according to Triton). My phosphate level is surprisingly low despite the increased fish load and extra feedings. Triton recommends feeding more and/or reducing the amount of phosphate remover used. As I don't use any remover that's going to be difficult to do but I do intend to feed more to my corals. Results comparison: Me Triton Ca: 410 434 Mg: 1272 1258 K: 390 386 Sr: 0-3 7.06 Iodide/Iodate: <0.01/0.03 0 PO4: undetectable 0.006 So what now? Well I'm going to continue performing water changes as normal, maybe change out a little more in the next a few weeks. Triton recommends 6 x 15%, but they are probably assuming that I don't perform water changes at all which is certainly not the case. I popped a polyfilter into the sump for good measure mainly because I had one to hand (from years ago, I hope they don't 'go off'?) and surely it can't hurt. I also intend to start dosing iodine again. Just before sending off the water sample to Triton as reported in a previous post, I also reduced the intensity of my lighting. It's been 14 days now and I do think that there has been an improvement in the corals, the colours look a tiny bit better and the polyps are starting to extend again. Perhaps the lighting was the main issue? I am hopeful that things will continue to improve. Now I just need to get rid of all that tin.... Photos to follow on Monday with the full May update.
0 Comments
Yesterday morning the male T. nudus goby was back out with the female again so I knew immediately that the eggs must have hatched at some time during the night. He had been caring for them non-stop for the last 7 days without eating so I was expecting something to happen within the next two days. I checked on the tank 2 hours after lights out the previous night but there was no sign of any fry at that point. When I checked again in the morning there were a few tiny goby shaped objects floating around the tank and one was even still wriggling but it was swept away by the flow before I could reach for a pipette to catch it. Unfortunately the fry don't really stand much of a chance of surviving intact by the time I get up in the morning to look for them. The powerheads are so fierce and the fry are so delicate, not to mention that I now have a shoal of hungry Cardinals that are always on the look out for any tasty morsels that happen to float by. I expect that they had a breakfast feast yesterday and the Acanthastrea looked suspiciously happy too!
At least I took better notes this time so I'm fairly confident now that the egg incubation period is 7 days so maybe next time if I stay up late or get up really, really early I can actually catch and photograph some..... The continuing saga of the pesky tiny Aiptasia.
I noticed the first tiny polyp growing out of the sand in the front left-hand side of the tank on the 22nd January, I immediatly siphoned it out with a pipette connected to some airline. Then on 5th March, two more appeared in the same area. They were promptly removed in the same manner. Unbelievably on the 10th March, yet another one appeared in the sand, what the?! Am I just removing the heads and they grow back from the base? Clearly siphoning them out was not working so this time I decided to nuke the little guy with some Aiptasia-X. Was that the end of it? Err no! A couple of days ago another one popped up in the same area. Arrrgggh! This time I took a scoop and removed the polyp and all the surrounding sand too. Only time will tell if I've beaten them this time, the strange thing is that they haven't appeared anywhere else in the tank... yet! Here it is in a glass cup, so I definitely removed something! Never in a million years did I think that it'd be easy to catch and remove my hitchhiking crab but boy how very wrong I was. My deluxe homemade crab trap actually worked first time! I propped an empty glass spice jar up against the top rock where the crab lived and baited it with some mysis shrimp just after lights out. I honestly felt that the jar was a little too tall for the job and it would never catch anything but to my very great surprise it did. I checked back on the tank after just an hour and was gobsmacked to discover the crab sitting in the jar. It's so great when a plan comes together without any blood being shed (and doesn't cost anything, lol).
With clearer photos I'm now confident that the crab is indeed Chlorodiella nigra. Please refer to the link below for comparitive purposes. www.crabdatabase.info/krabi/brachyura/eubrachyura/heterotremata/xanthoidea/xanthidae/chlorodiella/chlorodiella-nigra-2558 Of course I couldn't bring myself to kill the little guy, after all he had done no wrong, simply knocked a frag off the rock which was probably my fault for not fixing it down securely enough. The biggest issue for me was the fact that he'd set up home right next to my clam and whilst this species is reported to mainly eat algae I didn't want to take the risk of the clam being irritated or even worse becoming a crabby snack down the line. So off he went into the refugium where hopefully he'll be content with an endless supply of algae (and pods?) to munch on. ....probably, lol! After waiting absolutely ages for delivery I was finally able to set up my shiny new GHL Profilux 4 controller over Easter. The hope is that when the time comes I can monitor the tank whilst I'm away, so less worry for me. At the moment I just have it set up with four probes measuring temperature, pH, redox and conductivity. There is just 0.1 degree Celsius difference in temperature readings between the digital GHL probe and the Simplyaquaria temperature controller which is pretty good going considering the price difference between the two. I am going to keep the Simplyaquaria controller running as a backup to the GHL, two alarms are better than one in my books. I may actually move the Simplyaquaria probe to the DT so that I have two separate temperature readings to compare. The conductivity of the tank water measured by the GHL probe was 51.8mS which converts to a specific gravity of 1.0257. My calibrated refractometer measured it to be 1.026, again the values are pretty close which is good. I am happy to know that I've been maintaining a suitable salinity using the refractometer. I never bothered to purchase a pH test kit this time round so I had no idea what the actual pH readings were going to be. The levels are fluctuating between 8-8.2 or thereabouts, I would like to study the whole 24hr cycle but haven't managed to work out how to access data from the Profilux yet (if indeed it's actually possible to do). The redox probe is still bedding in, to be honest I'm not really sure what to expect from the readings as I've never used one before. It came with the set so I thought it'd be interesting to set it up and investigate what it can tell me about the tank. On the livestock front, Charlie the hitchhiking hermit crab is now on borrowed time after his first tank misdemeanor yesterday. He knocked my prize Acropora gomezi frag off the rock in his quest for food. I supposed I should be pleased that he just wanted to graze off the rocks and not eat the coral but still it's annoying. He's really quite big now so I'm sure that he'll cause more trouble in the future if I don't try and catch him now.
I think the T. nudus gobies have spawned again, Hop has been sequestered in the cave for the last two days and Skip won't let him out to feed, so I'm hopeful that there are eggs hidden inside. I hope I actually get to see some fry this time round, pretty please! The Cardinals also appear to be courting but there's been no further release of eggs. For a while now some of the corals have been losing colour. The worst affected being Acropora #3 (A. formosa?), the one that has been recovering from blistered skin. It's reached a point where some form of action needed to be taken. I had hoped that by introducing some more fish the nutrient levels would increase and the corals would look a bit happier, but even though I'm adding a lot more food to the tank the nitrate and phosphate levels still remain oddly undetectable. I performed as many tests as I could on Friday (with the exception of Iodine) and the results were as follows.
Specific gravity: 1.026 Ammonia: 0 Nitrite: 0 Nitrate: 0 Phosphate: 0 Alkalinity: 6.8dKH Calcium: 410ppm Magnesium: 1260ppm Potassium: 380ppm Strontium: 0-3ppm The levels are a touch lower than I'd really like them to be especially alkalinity and Strontium but the Tropic Marin Pro Reef salt mixes up to give an alkalinity level of 6.7dkH and 0-3ppm respectively so if I want those levels to increase I'm going to have to supplement them in my mixing bucket before doing a water change. I intend to add Reef-Roids to feed the corals more frequently as the filtration system I have in place seems able to cope with the extra loading. Along side that I also decided to alter my lighting schedule. I looked at some of the other LX7 lighting profiles posted on the GHL website and their levels were set lower than mine. Had I been unintentionally frying my corals with too much light I wonder? So now I have reduced the levels and the difference is quite noticeable to my eye, the tank looks a lot darker and quite a lot more bluer! To be honest I really don't like the look but if the corals respond positively then that's what counts. I expect I'll get used to it eventually. Daddy Cardinal made it all the way to the end of the 6th day mouth brooding the eggs and then on the morning of the 7th day they were gone. Soooo, did he get a bit peckish overnight and eat the eggs or did the fry hatch out and he released them?? I just don't know, it's really quite frustrating. First I missed the Nudus goby fry and now the Cardinals too.
Talking of the Nudus gobies their latest attempt at spawning (maybe) ended at day 4 thanks to the pistol shrimp who decided to fill in the entrance to their cave again. Following that disruption the gobies went MIA for 4 days and then both reappeared as normal. I wish that they'd just move over to the left hand rock pile and then they wouldn't be bothered by the shrimp's digging activities at all. My hitchhiking crab has been getting bolder and I've finally managed to snap a few photos of him. Charlie is much bigger now, at least 2 times larger than when I first spotted him or maybe even 3 times now! When I say 'him' I may be incorrect, in reality 'he' may be a 'she', so a Charlize as opposed to a Charles. Female crabs have a wide abdomen and males have a narrow one. In Charlie's case it looks sort of in between, an immature female perhaps?
Here's some pictures of the proud Father. It remains to be seen if he can carry the eggs for the full 7 days without eating them. Every now and again he will partially spit the eggs out, juggle them around a bit and then suck them back in again. It's really cool to watch.
I was just in the process of performing a water change when one of the Red Spot Cardinals spawned right in front of me!! The female released the most enormous packet of eggs where upon the male immediately took them into his mouth, it took several minutes for him to juggle them into position so that they all fit in. I really don't know how he did it, his mouth now looks fit to burst. To say that I was gobsmacked is a complete understatement, the fish have only been in the tank for six short days! This has to be one of the best days in my reef keeping journey so far.
I wished that I could have videoed the magical moment but I was busy siphoning water out of the DT, I'm surprised I didn't have a disaster whilst distracted. I will try to take a picture of the proud daddy in the next couple of days if I can, he's easy to spot with his bulging mouth. |
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|















 RSS Feed
RSS Feed
