|
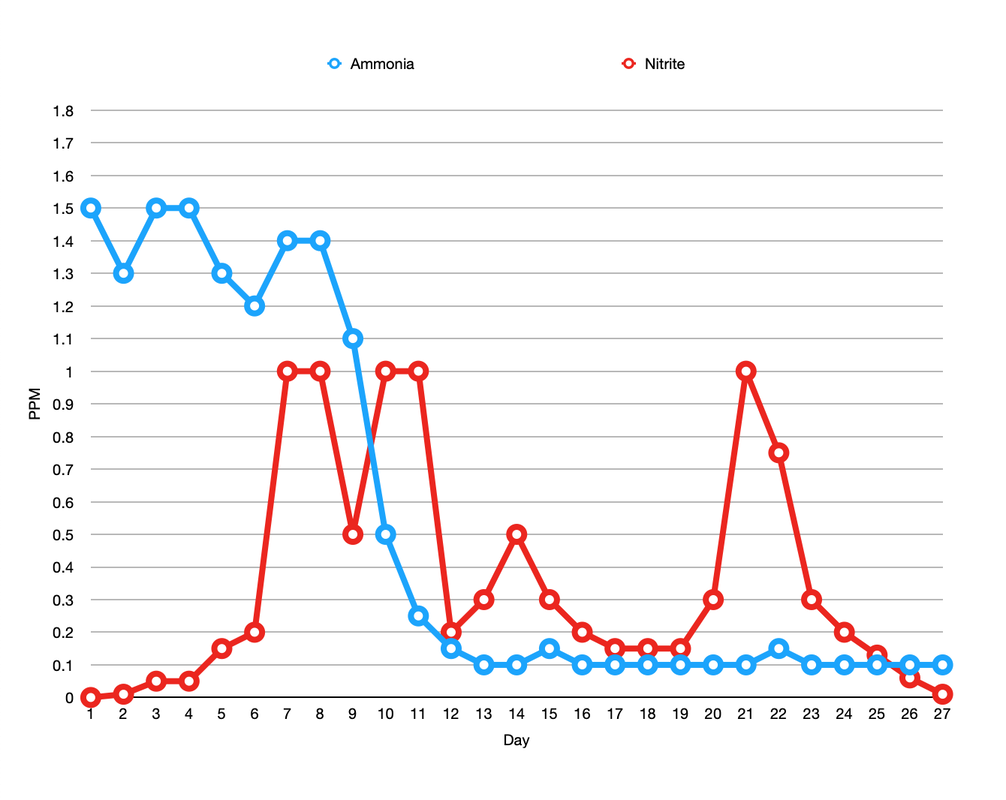
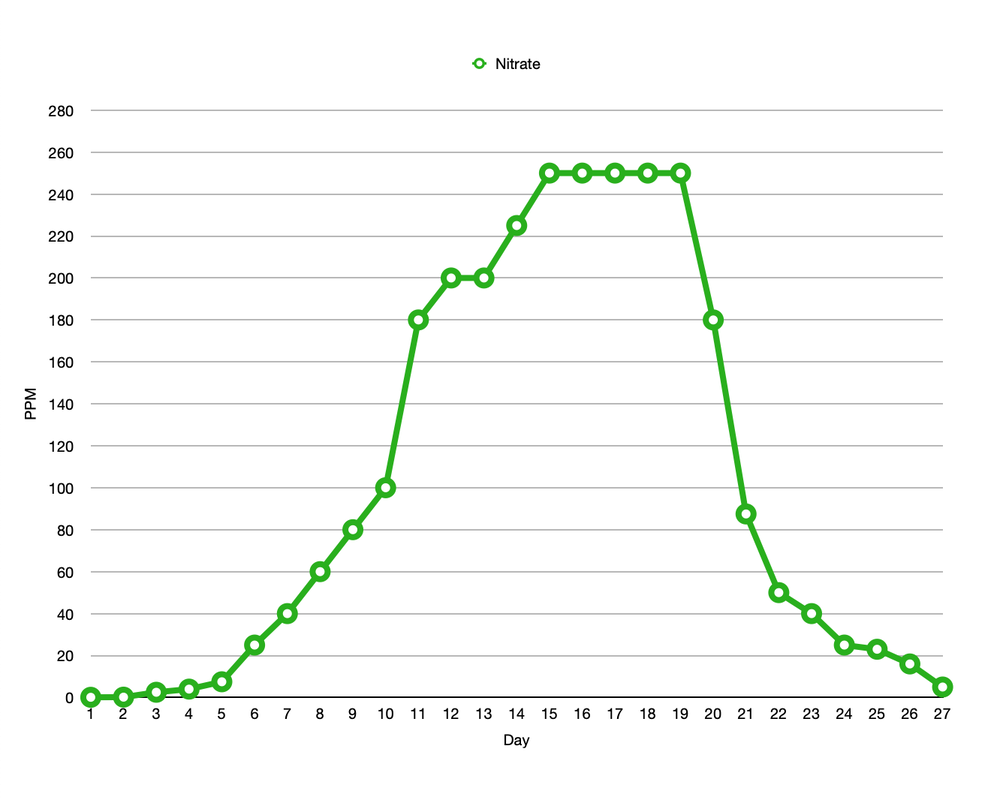
I began the tank cycle on the 2nd February 2020 with a bottle of Dr Tim's One and Only plus the recommended dose of ammonium chloride. Thereafter I tested ammonia, nitrite and nitrate every day for 27 days. Needless to say the levels I recorded in my tank did not look like the neat and tidy graphs I see posted online. I added further doses of ammonium chloride, as per instructions, on days 3 and 6. I also boosted the ammonia again on day 15 and 17 to keep things ticking over. The lights were left off during the cycle but there was some overspill from the Reefer 170. Here's what the ammonia and nitrite levels looked like. And here is nitrate. By day 10 the nitrate level had reached the upper limit of my Salifert test kit so I had to dilute the sample first by 1 in 5 and then later on by 1 in 10. On day 15 the nitrate level had reached a whopping 250ppm!! I envisioned having to perform water changes every week for the next 6 months or so. Things were starting to look a bit grim. The largest water change I can perform at any one time is 30% so on day 19 that's exactly what I did. The following day I recorded the level at 180ppm which is exactly the amount I expected following the water change but what I didn't expect was to see an increase in the nitrite level again. I continued my daily testing and not only did the nitrite levels fall back down again but the nitrate levels kept dropping and dropping till on day 27 they read a rather acceptable 5ppm with no further water changes required. How weird! I have a feeling that the off the scale nitrate levels were probably due to interference by nitrite, either that or I have a super duper nitrate reducing system in my tank. Patent pending. Whilst the tank was cycling my tank cover arrived and I was somewhat disappointed to discover that there is what I consider to be an excessively large gap between top of the weir comb and the mesh lid. What's that all about? Why can't the level of the weir be designed to be more in line with the top of the tank?? I don't have the D-D branded cover but I can't imagine that sits any lower than any of the other alternatives. Anyway there's more than enough room for any of my small to moderately sized fish to squeeze through and since there is no cover on the weir they'll end up stuck down inside it or take a ride down into the sump. Neither alternative is acceptable so I plugged the gap with some clear acrylic sheet. There is still a small gap to the right of the weir comb that I haven't fixed but it's pretty narrow so hopefully my fish won't discover it (famous last words I know). On day 11 I fired up the skimmer, an Ultra-Reef UKS-160 mainly for aeration purposes. This is not a new skimmer but you wouldn't know it, as soon as the pump was fired up it went completely crazy so I left the cup off for the time being. Plenty of air going in there to be sure. 10 days later it showed no signs of calming down and I was getting a bit sick of the salt creep everywhere so I fitted the cup with the drain plug removed so that the water overflowed into the cup and back into the sump again. As you can see protein scum began collecting inside the cup. Skip forward another 13 days and still the skimmer was in overdrive despite the flow restrictor being wide open and the unit sitting in exactly the right level of water. I decide to craft a stand to raise it up a bit, eggcrate, PVC pipe and cable ties later...ta da! That was version 1, since then the pipe legs have been cut down a bit as the skimmer was then sitting a bit too high (typical) but it's started working well now. I have read that skimmer collection is reduced in tanks where roller filters are also employed but it's definitely pulling out some stinky stuff. Talking of the X-filter, I must hold my hand up and admit how I eagerly watched and waited for the motor to turn for the very first time, yes I'm sad like that. It seemed to take an age for the water level to rise up and trigger the level sensor, lol. The excitement has since worn off a bit as I've become used to the motor just doing its thing, it is quite satisfying to see the used dirty brown roll. Better out than in (I hope). On day 27 I considered the tank to be pretty much cycled. Diatoms were beginning to take hold on the sand and rocks so I decided to add some Tisbe copepods. I wanted to encourage the growth of these little critters as much as possible before transferring any of (my pod loving) fish over from the Reefer. For a couple of days I thought the tank must be toxic and I'd killed them all off as I couldn't find a single pod on the glass but then they bit by bit they slowly started to appear. This made me one extremely happy reefer (I'm easily pleased clearly). On the same day as adding the copepods I fired up the refugium. I'm a big fan of refugia not just as an area for the growth of macro algae but also for the reproduction of beneficial critters (and also as a place to relegate any naughty crabs etc if needed). I wanted to keep the light spill down to a minimum inside the sump itself so I designed something to sit in the cupboard to the left of the sump with the pipework entering and exiting via the existing hole in the cabinet. I didn't want to have to modify the cabinet in any way and I almost, just almost, got away with it. The pipework was a tight fit but worked out perfectly, however the tank height was out by just over a millimetre, arghh! I had to sand down the back wall of the cabinet a touch to get it to fit. Ooops! I decided not to make the tank myself this time as it would be a lot bigger than my previous set up and hence more water to worry about leaking, instead I got Wharf Aquatics to make it for me. I'm running a Kessil H80 for the time being (that one I had lighting the Reefer refugium) but I may switch to something more viewer friendly at a later date, I'm not really a fan of the red/purple lighting to be honest. Here it is looking clean and tidy (needless to say it doesn't look like this now, far from it!). Incidentally even though I did not directly add any copepods to the refugium some have made it down there anyway from the DT and it's now buzzing with pod activity. I have the X-filter bypass controllers fully closed but still the pods have managed to find a way through the filter, past the skimmer and through the refugium pump/pipe and into the refugium, which I find pretty amazing to be honest.
0 Comments
Firstly another equipment change to report. I finally decided to change out the Beamswork EVO refugium light for a Kessil H80 Tuna flora. I had been considering replacing it for quite a while now, not because I was unhappy with the light but because the metal stand was rusting in all four corners where it sits on the top of the refugium. I'm not sure if this has caused any metal contamination in the tank yet or not but I wasn't happy with the situation so it had to go. The reason I opted for the Kessil is because it's pretty much the only light that I could find small enough to fit over the refugium, everything else was either too big/long and would have caused a huge light overspill issue or excess heat in the small sump area. To combat at least some of the light spill produced by the H80 I have added a small piece of black acrylic sheet between it and rest of the sump. The red light is something that does take a bit of getting used to but if the macro algae grows well under it then I'm happy, time will tell. Now for a Fauna Marin Skimbreeze update. The FM media lasted two and a half weeks before needing to be replaced, about the same amount of time as the most recent ATI 1l disposable unit. I'm not that keen on the design of this reactor itself as air is preferentially pulled from the side the outlet tube sits (as can be seen in the photo below). So when changing out the reactor some media is still unused. I think I will try rotating the cylinder next time to see if I can reduce the problem. To refill the reactor I used the Spherasorb media, it looks exactly the same as the FM stuff but costs a lot less. As for the livestock well everything is ticking along. Acropora growth is slow to nonexistent thanks to the red bugs but at least they are not dying. The exception to this is the A. hyacinthus which seems fine (with a rather odd growth shape it must be said), I've accidentally fragged it with the algae magnet a couple of times now because it's growing too close to the front glass. The Utter chaos zoanthids are becoming annoying as they keep detaching and floating around the tank ending up in spots where I don't want them in or sucked on to the powerheads. Since I don't have anywhere to place frags (and hate the look of frag racks in the DT) I've chucked the loose ones I've found so far. The sun coral is still releasing planulae and I keep finding baby polyps popping all over the tank. The more I look the more I can spot, they've even settled out on the clam shell. The larvae are fascinating to watch, tiny orange tear shaped blobs that wiggle around the tank until they find somewhere to settle out. It's very cool! As for the Balanophyllia now living down in the sump, I can't decide if it's doing better or worse. There is one thing though, I happened to knock it over whilst trying to feed it and when I set it straight again I noticed it has some tentacles poking out the back, my initial response was Oh ****! Aiptasia!! But on closer inspection it actually looks like a baby Balano bud. I'm not sure if this a good sign or a bad one? Is it failing so badly that it's sending out buds as a last ditch attempt to survive?? I surely hope not. I'll sign off with a few photos. And finally a FTS..
|
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|







































 RSS Feed
RSS Feed
