|
It's been a while since I posted a fish update. I've been putting it off because it doesn't make for good reading. I lost my Helfrichs firefish in September last year, I'd kept him for just over a year. There was no obvious cause, he just ate less and less until he eventually passed away. At the time I put it down to just one of those things. All was well for the next 4 months but then my Rainfords goby started to go downhill in the same manner, he began eating less and less till he too died at the end of January. I'd kept him for just under 2.5 years. He was a big fish, almost certainly fully grown and perhaps it was just his time, but still I did wonder if the bullying by Yellow wrasse was somehow related. There wasn't any actual fighting just a bit of posturing every now and again but it might have been extra stress that he could have done without. Then three weeks later I also lost my beloved Pink Streaked wrasse. I'd kept her for just over 4 years, according to information found on the internet the average lifespan of these small wrasse is 4-5 years so it's quite likely that she did just die from old age. The following month I lost one of the pair of Blackbarred gobies, it too stopped eating and died. Just 6 months with me for that fish. Then in April I lost the Fathead anthias (after 13 months) and in May my beautiful Spotted Mandarin also died (just under 2 years with me). When I removed the Mandarin's body it looked perfect in every way, nice and fat with no torn fins.
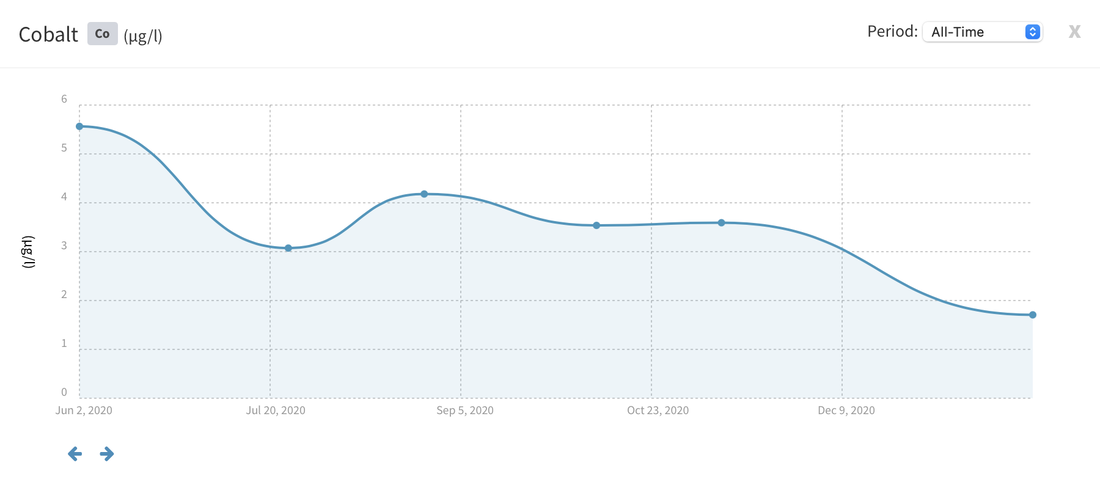
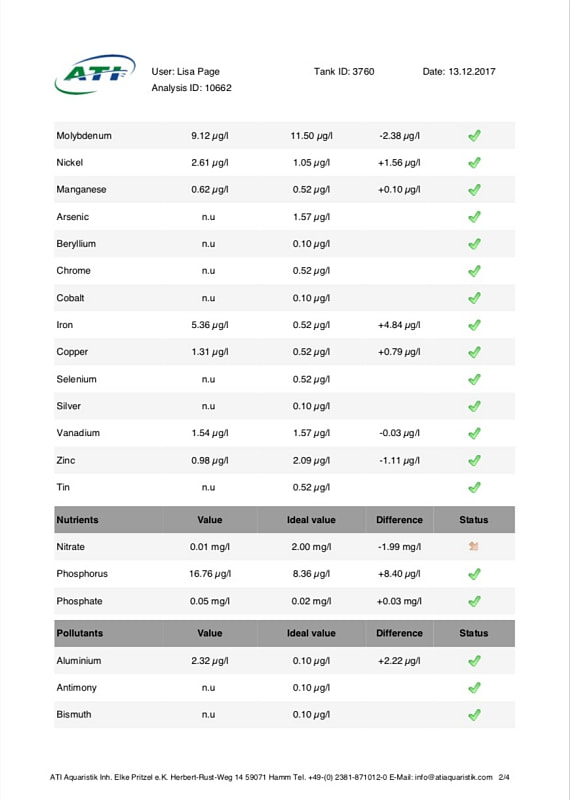
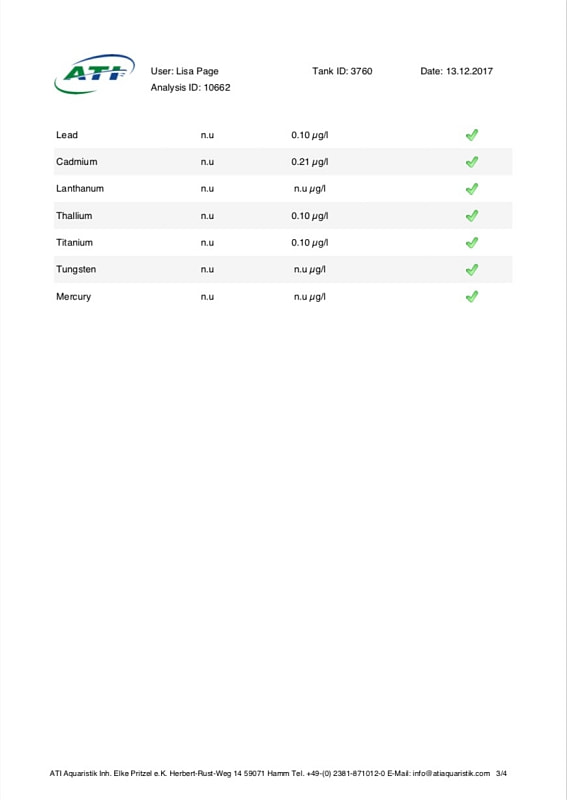
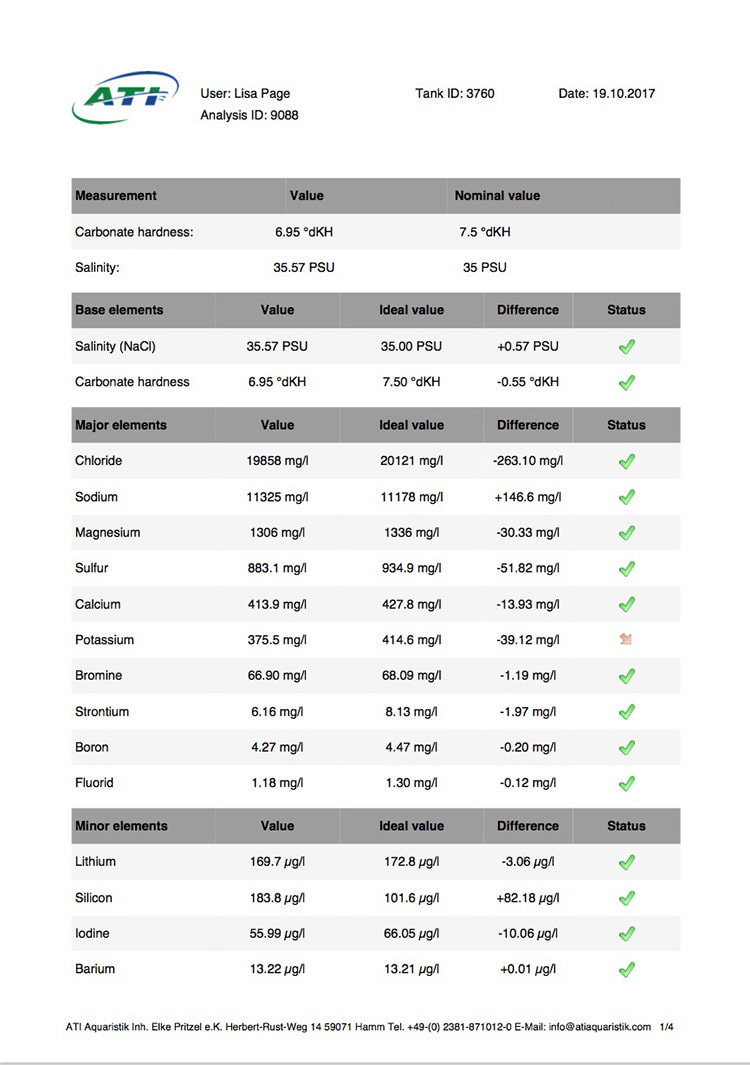
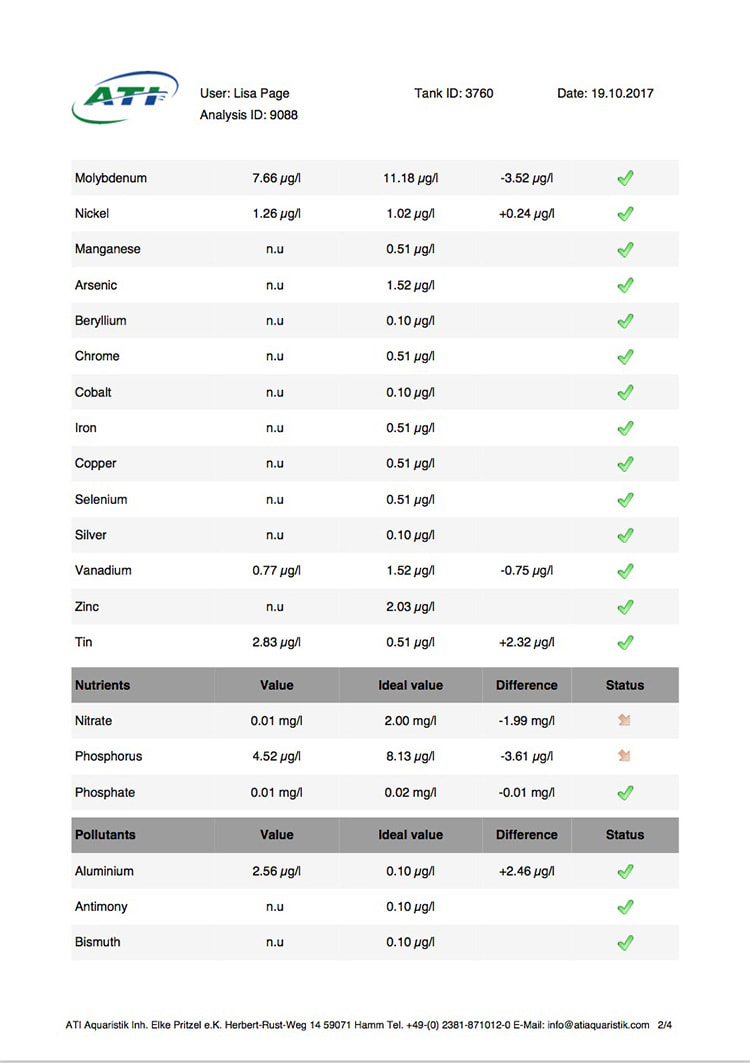
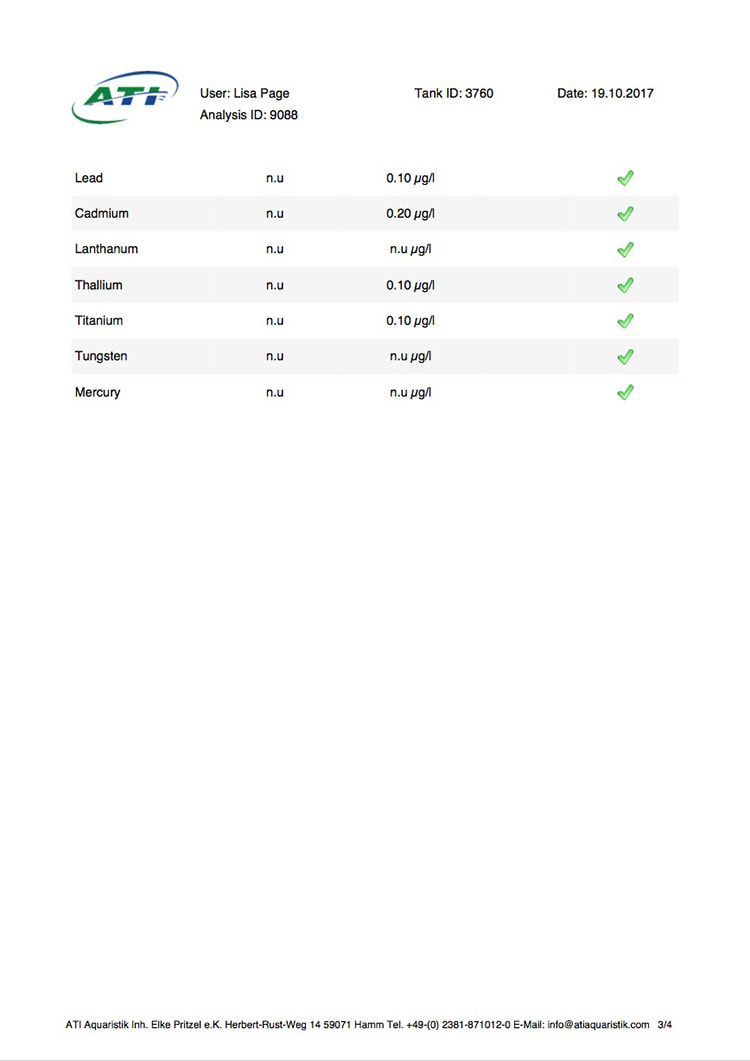
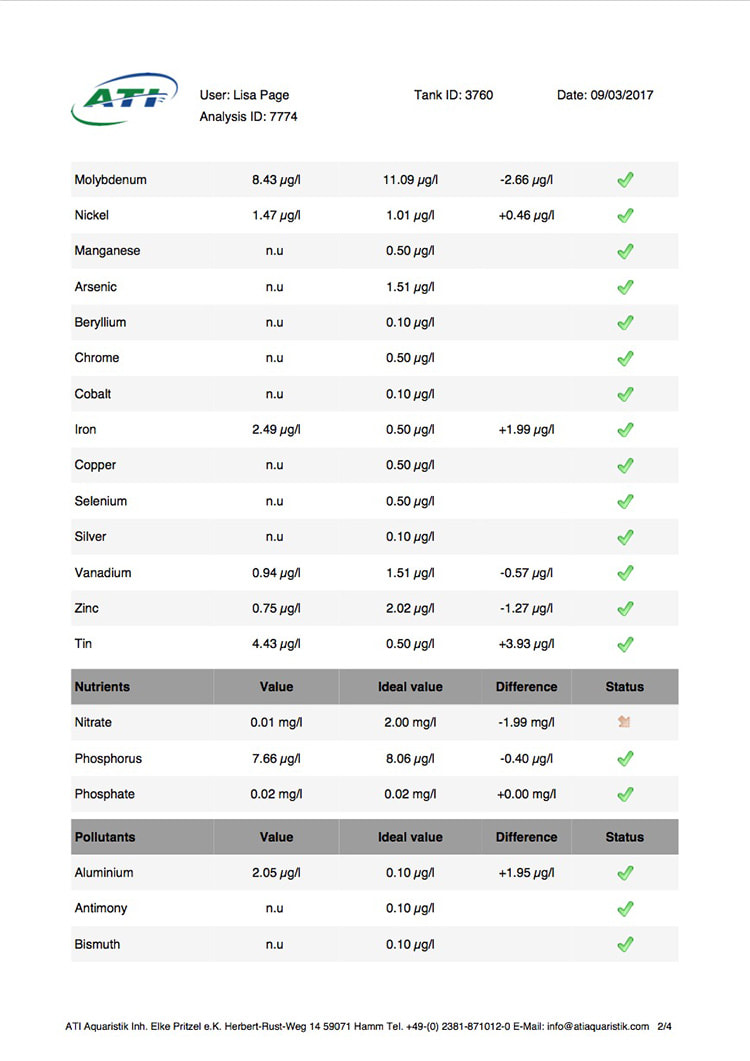
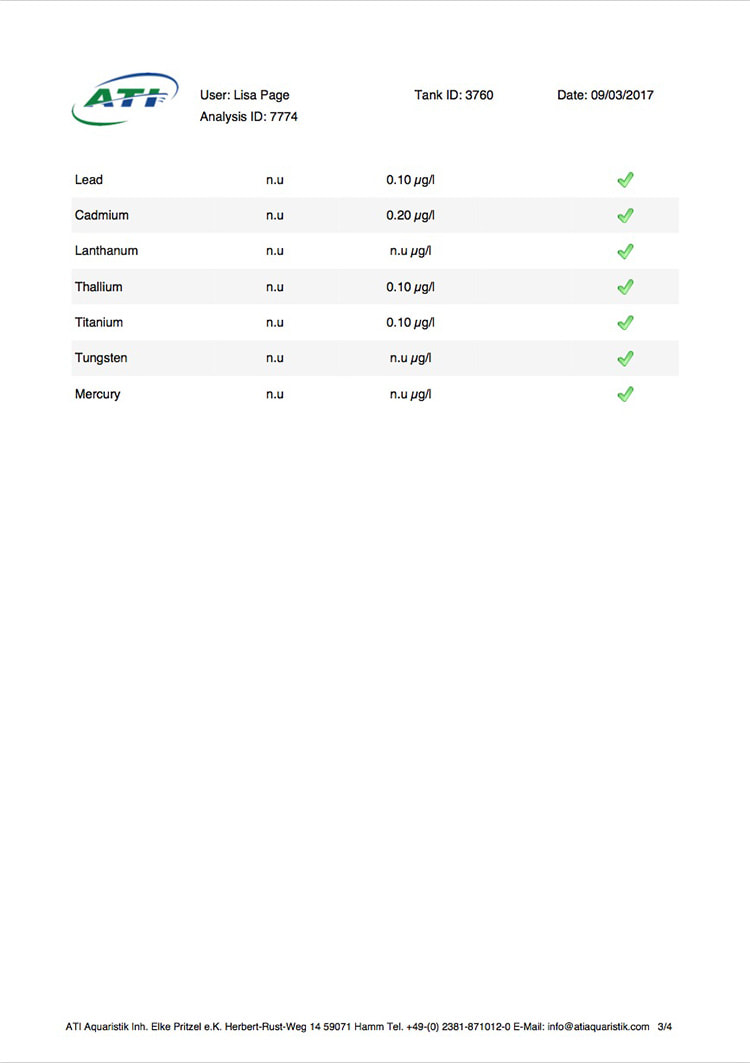
Now I don't really know what to think, maybe the deaths are totally unrelated but I am beginning to wonder if there is something in the tank silently killing fish in my tank. I sent off for an ICP analysis at the beginning of May and everything seems fine except for Cobalt which is still elevated. 15 months and the TMC Ecoreef rock is still leeching cobalt!! I hate this rock so much, it's hands down the worst decision I have ever made in all my years of reefing. Looking at all the test results, it's going to be many more months, maybe even another year before the cobalt is finally gone. I have scoured the web for information about chronic cobalt poisoning but haven't really turned up anything useful. Most research involves short term exposure of fish to much higher levels. lab.atiaquaristik.com/share/7132f6e0d9425dd6e201 It goes without saying that I am hesitant to introduce any new fish to this tank now. At least the corals appear OK, if they were slowly dying too then I'd be ripping out the rock and starting afresh. My current fish list is: 1 x Tanaka Possum wrasse (4.5 years) 1 x Yellow wrasse (3 years 9 months) 1 x Whitecap goby (1 year 7 months) 1 x Blackbarred goby (9 months) 5 x Ghost cardinals (6 months)
1 Comment
Yesterday I received the results of my latest ICP analysis and I'm relieved to say that the cobalt level has dropped to 1.71ug/l. It's still 17 times above the reference value so there is still a way to go before it's gone completely. I wonder if the drop is the reason for the recent turnaround of my Acropora hyacinthus?
Link to the latest ICP: //lab.atiaquaristik.com/share/5c6ace3e71c6dbce4bea Graph showing the ICP cobalt levels since the first test, the rock had been in the tank for 5 months at that point. It's now one week shy of being a year old. I am generally a patient person but after almost ten months of waiting my patience is wearing thin. ICP analysis has indicated that the tank still has an issue with elevated Cobalt levels. Having discussed my results with other reef tank owners the source of the problem appears to be TMC Eco-reef rock, some have even had it a lot worse than myself, perhaps because they used more rock?
I have sent 5 sample of water off for testing and the Cobalt level has come back as follows: May: 5.56ug/l July: 3.07 August: 4.18 October: 3.54 November: 3.59 I have performed 12% water changes every week without fail since the tank was cycled. If the rock had stopped releasing Cobalt or was at least releasing less I'd expect to see the level dropping. It's hard to tell if or what kind of negative impact the Cobalt could be having on the livestock. I am still struggling with algae issues on the sand, this could be down to all sorts of reasons but I never had an issue with my previous tank set up(s) using good old liverock. The KH level has also begun to rise recently indicating that the corals have stopped growing for some reason. The Red Planet Acropora, A. hyacinthus looks to be holding on by a thread at the moment, it's as brown as I've ever seen it with little to no polyp extension. I get up every morning expecting to find just a stripped skeleton. I also lost Milo my Venus shrimp recently which I am absolutely gutted about. He was completely fine one day, stealing food from the Heliofungia and just gone the next. I have no idea what happened to him, was it a failed moult? His death may be completely unrelated to the Cobalt levels and it could be just one of those things. I really hate not knowing what happened to him. I received the results of my second ICP analysis yesterday. Cobalt and Aluminium are still elevated but there is a little less than before so maybe the TMC eco rock has stopped leeching or is a least leeching less now, time will tell. The nutrient levels are still low despite my feeding a ton of food and dosing extra nitrate. I have been seeing a little bit of green cyano on the rocks and the sand is looking a bit greener too. I'm still debating whether I should do anything about it or just wait and see. I'll probably just wait and see. Oh and I have been plucking out tiny bits of Ulva from the sand (mainly) whenever I see them, grrr! I'm resigned to have to keep doing this from now on. The sun coral has christened the new rock, the first baby is coming along nicely. The baby Trochus snails are doing really well. I've been moving any that I find in the sump over to the refugium. The refugium needs cleaning and they are better off in there, I want to avoid the scenario where they get crushed by an impeller or jam up my X-filter. I have discovered some in the DT too which I'm surprised about, I thought that they would all have been wrasse food but I guess they are able to hide well enough to avoid such a fate. They do blend in with the rockwork extremely well. The Coco worm continues to do surprisingly well, it has extended its tube even more now. Kylie the Pink Streaked wrasse is keeping a beady eye on it for me. I'm also pleased to report that the Menella gorgonian appears to be doing great now. It's finally decided to pop out another branch at the base, woo hoo! Unfortunately I can't get it to completely recover the sections that lost some flesh earlier on because hair algae has taken a hold. It's only possible to see the algae when the polyps are fully retracted but it does annoy me no end. When the algae grows long enough a hermit comes along and gives it a trim which I appreciate. Unfortunately the Rei Yellow wrasse seems to have taken a bit of a disliking to Jessie the Rainford's goby, I have no idea as to why; a dominance thing maybe. There is no chasing or actual fighting but a fair amount of posturing goes on between the two. When they meet they both fully flare their fins and engage in some sort of a staring contest, Jessie may be the smaller fish but he stands his ground. Rei had better watch his step because if it comes to a choice he'll be the one to go. Catching him would be the issue... A quick pic of Sunny the Sunburst Anthias.
I let the tank and myself have a breather for a week following 'clamgate'. Clive the clam appears to be fine after the incident and I'm pleased to report that nothing succumbed to an excess of clam ‘protein'. Then I transferred over my two remaining Acropora (gomezi and hyacinthus). I'd been putting off moving these because as we all know SPS can be tricky in new systems but it had to be done and if they didn't make it that'll be that. The A. gomezi was super easy because it's a tiny frag on a frag plug but the A. hyacinthus had grown rather large. I tried my best to frag it off the rock in one complete piece but, yeah that didn't happen. It broke into three, I was going to keep the two largest but in the end I opted to add a single bit only (easier to fix down). Those who followed my old thread may remember that the Reefer 170 had an issue with red bugs. I hadn't seen any of the little red devils for ages but that doesn’t mean that they were all gone, I can only hope. I dipped both frags in Reef Primer and checked them quite thoroughly before they were transferred.
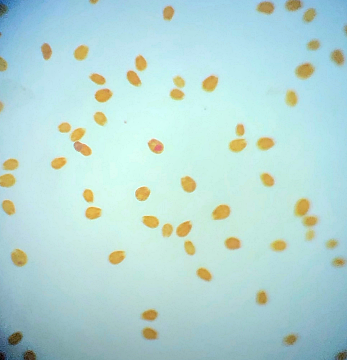
Now we get to the big one. On the 26th April I decided to complete the tank transfer and shut the Reefer down. Prior to this I made one last concerted effort to catch Rei the yellow wrasse and Spike firefish but Rei would only poke his head briefly into the trap and Spike avoided it entirely so it was simply a no go. We did try catching the Spike using nets and acrylic baffles, much chaos ensued with the tiny fish outwitting us every time. The strip down progressed pretty much as planned with no disasters to speak of, I found it quite stressful nonetheless. We emptied out the water and rocks in stages. The corals that I wanted to keep were cut off and placed in one bucket with the remaining rocks/corals going into another. Spike the firefish was cornered with a net and safely removed. Ming the Pom pom crab was discovered clinging to the underside of a rock. Finally we were left with a tank containing a little water, one last piece of rock and sand. With my breath held, the rock was lifted out revealing Al the pistol shrimp and Flash his Whitecap goby partner hiding underneath. Phew! A careful bit of sand exploration was required to flush out Rei the yellow wrasse. Swipes the porcelain crab was MIA at that point so we had to carefully go back and examine every bit of rock again placing them one by one back into the Reefer as we went. Finally we discovered her hidden in a hole in one of the base rocks. That was pretty much it except for one last thing of note, I discovered a rather scary number of Aiptasia living in the overflow weir along with half a dozen baby sun corals. Following the transfer Rei the yellow wrasse hid in the sand for a whole 10 days before finally deciding to make an appearance. I honestly thought he’d died from stress or something. Everything else made it through OK which I’m relieved about. The fish were naturally pretty freaked out however so I didn’t take any photos for ages. The Acros are still alive and growing but have lost colour which is probably down to the poor nutrient situation. There have been a number of reports circulating recently that TMC eco reef rock leaches phosphate and silicates but apart from a brief spike of phosphate during the cycle that hasn’t been my experience at all. Nitrate and phosphate have been consistently registering as zero on my test kits (Salifert and Hanna respectively). In the old days this wouldn’t have bothered me much but the internet has taught me to fear the dreaded D word. For a while I saw a little growth of what looked like some brown algae on the rocks but when viewed along the length of the tank with natural lighting behind was in fact green hair algae. There must have been some nutrients knocking about somewhere to fuel the growth. This algae started to become a little more pronounced so I decided the CUC needed a tiny boost. Two weeks and 6 small Trochus snails later most of this algae was gone, I felt quite pleased with myself. However the removal of the hair algae shifted the balance somehow and combined with a lack of nutrients I began to see the appearance of dinoflagellates on some of my gorgonians. Normally I would take a watch and wait approach but it’s hard not be affected by some of the algae horror stories I have read online. I dusted off the microscope and identified the species as Ostreopsis, fearing a full blown infestation along with the death of my beloved snails I decided action was required in the form of nitrate dosing. I had already been feeding quite heavily to that point including Reef roids and phytoplankton but it didn’t seem to have made any measurable difference. It felt wrong to be actually dosing nitrate! I began with a laughably tiny amount (0.5ml of Brightwell’s NeoNitro per day, 12.6ml will raise my tank volume by 1ppm so 0.5ml was nothing lol). After 6 weeks I gradually increased the dose to 4ml per day, during this time the dino growth increased slightly, mainly evident on the gorgonians, but never turned into the major disaster I feared. The gorgonians still had their polyps extended and the snails seemed fine too. I continued my weekly water change regimen using the opportunity to siphon as much of the dinos as possible every time (not recommended I know but I like to perform water changes). When the tank was 4 months old I sent off the first ICP sample. I discovered elevated levels of Cobalt and Aluminium. I have no idea where they have come from, leeching from the rock maybe? The nitrate level was 0.02mg/l, phosphate was 0.03mg/l and silicate was 66ug/l. Iodine was a bit low as expected (and some other elements low as per usual). //lab.atiaquaristik.com/share/3023c74a1cf286bcd614 Finally after 7 weeks of dosing nitrate I started to register 1ppm on the Salifert kit, incredibly the dinos started to recede! This might have been a coincidence of course and nothing at all to do with the nitrate level but either way I am happy. I am curious to know why I’m not registering a release of phosphate and/or silicate from the TMC eco reef rock. Perhaps the corals are soaking up the nutrients as they are released, the gorgonians are growing very fast. Or maybe the rock is hatching a plan to trip me up later on down the road. I have lots to update, hmm where to begin? I think I should probably get the bad stuff out of the way first. Forgive me Father for I have sinned. For quite a while I had noticed that I was harvesting less and less macro algae from the refugium, growth had slowed to almost nothing at all despite there being plenty of nitrate and phosphate available. The days of my tank having low nutrients were certainly in the distant past as they had been slowly but surely creeping up. At the last ICP analysis nitrate was sitting at 11.5mg/l and phosphate at 0.13mg/l. I'd also noted that the mini brittle stars that used to thrive in amongst the algae had dwindled from hundreds to zero (there are still lots in the DT). I'd come to the conclusion that Charlize the hitchhiker crab had been supplementing her diet with some brittle star meat. Anyway hair algae had taken a firm hold in the upper half of the refugium and was choking out the growth of macro algae below. This meant that there was even less algae available to the crustacean residents (Charlize and also Bruce the Emerald crab) at the bottom of the refugium. Then at the beginning of December, I discovered what was left of Bruce, had he also been eaten by Charlize? RIP Bruce. So on the 16th December 2019 I decided then to take the refugium out for a good clean, removing the old mud substrate and replacing it with some new, a long overdue task I must admit. I carefully salvaged as much of the macro algae as I could (a mix of Caulerpa racemosa and Chaetomorpha) and placed it, along with the naughty Charlize, into a bucket (with tank water obviously). I then disconnected the refugium and set about cleaning it. Once everything was clean(ish) I added a new layer of mud, refitted the refugium and carefully filled with water. After a while I reintroduced the macro algae and crab. So far so good, however things were about to take an unfortunate downward turn. It seems that by cleaning the refugium and/or replacing the mud I had altered the water chemistry and not in a good way either. The redox value dropped to 250mV and stayed there. Under normal circumstances the redox probe reads somewhere between 350mV to 450mV and to be honest I don't pay that much attention to it. The numbers bounce around depending on whether I've just fed the fish or done a water change. A value of 250mV however was definitely not normal. I wondered if the probe was reading accurately so I cleaned and recalibrated it but still the readings remained low. A few days after cleaning I noticed some suspicious looking brown algae starting to appear in the refugium and I just knew this wasn't going to be good. Sure enough after about a week or so I started to see signs of it appear in the DT too. It began by coating the gorgonians, the Plexaurella was particularly affected and closed up. Ten days after cleaning I discovered Charlize the hitchhiking crab dead in the refugium. Nooo! I couldn't believe it and felt so terrible guilty. Why did she die? My immediate thought was that the brown algae may have been the cause as some species of dinoflagellates are know to be toxic. I've have not been unfortunate enough to have to deal with this type of algae before but I've certainly read about it a lot. It looked just like typical dinoflagellates, brown and snotty with trapped air bubbles but just to be sure I took a sample and dusted off the microscope. My suspicions sadly proved correct. The tiny oval protozoans were swimming in a circular motion around an anchor point like a tetherball which is typical of Ostreopsis sp., this is indeed toxic to snails and other herbivorous creatures. I've read many horror stories regarding dinoflagellates in reef tanks, so to say I was feeling depressed was a bit of an understatement, I had visions of all my corals covered with brown snot and the sand littered with shells of dead snails.
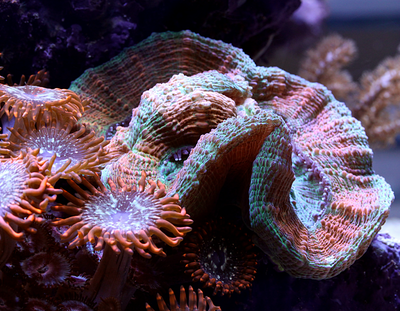
According to my research there is no easy way to rid a tank of dinoflagellates and I certainly wasn't keen on the idea of performing a 5-7 day black out. I decided not to panic and continue tank maintenance as normal. I continued with the weekly water changes (yes I know these were not advised) and siphon out as much of the 'snot' as possible in an effort to give the gorgonians a tiny bit of relief. I don't know if it helped them but it certainly made me feel better. In the ensuing days the dinos spread to the tips of the Seriatopora hystrix but surprisingly nothing else seemed affected. Every morning I would count the number of snails to make sure they were all still present and correct. The redox level remained very low so on the 14th January I sent off a sample of water for ICP analysis just to check if the mud was leeching out something nasty but as you can see from the link below the results looked OK. Nitrates and phosphates were lower than the previous test but not that low. lab.atiaquaristik.com/share/6bba53665864d463b982 Since I'd effectively removed most of the beneficial bacteria and critters from the refugium (except for amphipods and mysid shrimps) I decided it might be a good idea to add some diversity back in the form of some live rock rubble. This proved easier said than done as nobody seems to stock actual live rock anymore, it's all artificial or dead rock and bottled bacteria these days. I tried ordering some in from a local shop but when I went to collect it, it was just a bag of dry rock, sigh! In the end I located an online shop that was out of stock but expecting a fresh delivery of Australian live rock in the next few weeks or so. I decided to preorder a small amount and crossed my fingers that it wouldn't take too long. In the meantime the redox gradually started to creep back up again and by the end of January it had hit 350mV once more, the gorgonians started to pick up again. The first to bounce back was the Muricea followed by the Pinnigorgia and finally after over a month of looking completely dead the Plexaurella shed a layer of algae/mucus and the polyps came back out again, sadly a couple of branches had stripped but at least some of it was still alive. I also only had to frag off one of the Seriatopora branch tips and the rest bounced back in no time at all. As of today the dinoflagellates have almost completely disappeared from the DT, if you check the Pinnigorgia very closely there is still some evidence of some thin brown strings in one area of low flow but there's not much left at all. I am hoping in another month or so it will have gone altogether, I have my fingers crossed. Oh and just as things were starting to look up I received acknowledgement that my small order of live rock was available and ready for delivery (20th February 2019). That story will have to wait until another post, oh the stress is never ending.... First the not so good news, I think it's time to hold my hands up and admit defeat when it comes to growing Acropora in this tank. As soon I discovered the first red bug I feared the worst and that certainly appears to have been the case. I had hoped that the corals would be able to survive the parasitisation and for a while they did, growth was slow but steady and colouration looked OK but that's certainly not the case now. To recap, patient zero (the frag that initially infected the tank) was removed leaving a small amount of encrusted base in the tank, it looked like was going to recover and regrow but progress was so slow that the neighbouring Montipora (Superman) completely overgrew it. The second casualty was the Purple-tipped Acro, this suffered from STN (slow tissue necrosis) and had to be fragged. The remaining frags encrusted onto the rockwork but again showed little upward growth and were in time also overgrown by the Superman Montipora. A tiny bit of encrusted base still remains but I fully expect it to fade away or be overgrown eventually. The A. loripes suffered from RTN (rapid tissue necrosis) whilst I was on holiday and the Red Dragon joined it shortly after. The A. echinata frag also suffered from STN and was fragged but the stress of it being chopped up caused the remaining branch to strip overnight. So this leaves me with Acro #1 which has browned out and is soon going be completely overshadowed by the green plating Montipora (I never actually wanted any plating Montipora in this tank but this was a freebie coral that came in along with an order of encrusting forms and I, like an idiot, assumed it was encrusting one too, duh!). I also still have Acro #3 which is struggling for space next to the large gorgonian and A. gomezi. At first glance the A. gomezi looks OK but is actually covered with red bugs and has put in no upwards growth since its introduction almost 18 months ago!! There is one Acro however which appears unaffected by the Red bugs and that is the A. hyacinthus (aka Red Planet), this has grown relatively well and in fact has to be fragged because it keeps growing too close to the front glass. I cannot find any trace of red bugs on this coral. So there you go, it's no more Acropora for me, perhaps I'll be able to restock one day when the remaining Acros have gone and the red bugs have died out without a coral on which to host. In case there was something going on with the water quality that I wasn't aware of, I did send off for an ICP analysis but nothing stood out as being particularly troublesome. Strontium, Barium and Manganese were on the low side. Nitrate and phosphate were 3.02mg/l and 0.09mg/l respectively which is the highest they have been but not excessively high, I think (maybe someone may correct me?). Still, I have decided to run a little Rowaphos to bring the phosphate level down a touch. Full results can be seen via the link below. lab.atiaquaristik.com//share/b48ad381479dbd88fe1c Now for the better news the rest of the livestock for the most part appears to be doing fine. The Stylophora, Montipora and Seriatopora are all growing great (a bit too great actually). The lovely beach bum Montipora is thankfully still with me and finally showing signs of growth too. Unfortunately it's becoming harder and harder to see with the ever increasing Stylophora situated to the left of it and monster green plating Montipora to the front. Very poor planning on my part! I can just about catch a glimpse of it from the top but I suspect that won't last long. I would really love to move it to somewhere viewable but I doubt that I could get it off the rock even if I had somewhere to move it to which I certainly don't. The Superman Montipora has completely filled in the space I gave it and is also growing up the side of the Crocea clam shell. I'm not sure if I should be worried about this development or not. I hope it doesn't impede the opening and closing of the clam. I had to remove the original frag of the Cyphastrea as the Favia next door was remorselessly attacking it every night and anyway I never liked the look of the horrible frag plug. Since its removal the remaining encrusted part has been doing much better, it's still very close to the Favia but seems to have fallen under the radar for the time being. As for the Lobophyllia I had always been led to believe that they are super aggressive corals but the Utter Chaos zoanthids are creeping ever closer without any form of retaliation at all. I had rather hoped that it would keep them in their place. Sadly the Sunrise Goniopora frag is not doing very well at all. To begin with, it looked great and was encrusting on to the rock it was placed but then it started to lose colour and not extend as much. I tried moving it to the back of the tank where it was less bright but that didn't seem to make a difference. I can only assume that it hasn't been finding enough food, I do target feed it Reef Roids and Goniopower but generally not more than once a week. I am ever conscious of the size of the tank and nutrient levels. Here are a few recent photos: Now on to the new stuff! With the loss of a few of the SPS corals I was needing a pick me up and a new coral (or two as it happens) does the job. For ages I had fancied adding a short tentacled Fungia to the tank and since we had a trip to London planned a couple of weeks ago, a visit to Advanced Aquarium Consultancy was in order. I couldn't believe my luck when I found out the night before that they were having a sale on some of their corals. I thought "fantastic", I can save some money for a change, lol! However instead of saving money I spent more as I ended up buying a Scolymia instead of a Fungia. Oops! It's funny how that happens when shopping for corals. Needless to say I could have spent more, waaayy more, thank goodness I live too far away for AAC to be my LFS. Apparently it's a Reverse Bleeding Apple Scoly (more red colouration than green) and here it is after 4 days. I've been watching it like a hawk, worrying about its health because frankly it's the most I've ever paid for a coral even with 20% off. Oh but it's so pretty. I had to remove 4 Asterina sp. starfish from the base of the coral after acclimation, I felt bad about killing them but really, I have enough pest issues to deal with as it is. It's a shame because close up they are quite nice to look at and I love starfish but I can't take the chance that they may become a nuisance. Although it would be cool to have a Harlequin shrimp... Here is the Scoly in night feeding mode. Just after the addition of the Scolymia I discovered an online seller was offering frags of Dendrophyllia sp. Now I really love sun corals (well, non-photosynthetic corals in general) and my current Tubastrea is doing great so I convinced myself that I could squeeze in an itty bitty frag of a Dendro too. The coral was listed as yellow/red in colour with the polyps opening up in the day time as well as the night. However as you can see the coral is a uniform lemon yellow in colour with not a hint of red at all (unlike the one shown on the website). No matter though it looks healthy and really rather lovely, it opened up much faster than I expected and is taking in food already which is excellent. I can confirm in my vast experience of one week that it does indeed stay open during the day. I'm not convinced that it's a Dendrophyllia sp. however, it could alternatively be a branching Tubastrea sp. Only an examination of the skeleton will determine that for sure and since I'd really rather it didn't die I'll be content without a positive ID. Here's a full tank shot to end this update.
Just prior to the skimmer failure I sent off water samples for yet another ICP analysis. The results of which are shown in the link below: 100%! Apparently I have a full house, everything is in balance although to be honest the salinity is a bit higher than I would like. I must try harder. The conductivity probe had drifted out of calibration. The tin contamination is gone at last but I seem to have a reading for iron this time round. The only change between now and the last test is that I have begun to soak the fish food in a vitamin supplement, I wonder if that’s the source? Something (else) to keep an eye on. I still can’t get much of a nitrate reading despite adding a couple more fish and feeding loads. I wonder if running the tank for 5 days without a skimmer has raised it at all? Moving on to the corals, the warfare continues. The Favia continues to batter the Cyphastrea relentlessly. I thought it might stop once the leading edge was dead but no, the dead area seems to get larger every day. I would move the Cyphastrea if I could but firstly, it's well encrusted, and secondly I have nowhere else to put it. Two of the Montipora sp. (#2 & 3) are now clashing with Acropora loripes and both are losing. Montipora #2 is also being hammered at the back by the Stylopora. Despite religious feeding the Balanophyllia has still not been doing so well, It was only after I made the decision to move it that I discovered that it was receding very badly at the back where I couldn’t see. There was a white band of what I assume to be a bacterial infection at the receding edge. I relocated it to the rear of the tank but in hindsight I should probably have placed it in the sump because as soon as the Pintail wrasse were added, feeding it became nigh on impossible. They just kept stealing its food. Fortunately the Sun coral continues to do well and has grown multiple new heads. Feeding it can be a battle with the wrasse but it can be done with a little perseverance. Moving on to the red bug problem, with no plan of attack they are obviously still present but the Acros seem to be coping with them for the time being. I fully expected the afflicted corals to be failing by now but they still have reasonable polyp extension and colour. I'm sure that they could look better as could their growth rate but at least they are not dead. The watching and waiting continues. I've indicated some of the red bugs present on the Acropora below with red arrows there are more shown in the shot but you get the general idea. As for the fish, Edna the Possum wrasse passed her first anniversary in the tank on December 18th and Kylie the Pink-streaked wrasse will have her first anniversary on the 28th January 2018. A few more photos of the Pintails (Tinker and Belle) and Rei the Yellow wrasse too. A few other random coral photos. I had hoped to have more shots to share but Christmas preparations got in the way. Thanks for following my blog, I hope you all have a great Christmas! Hopefully I have more photos to share next week.
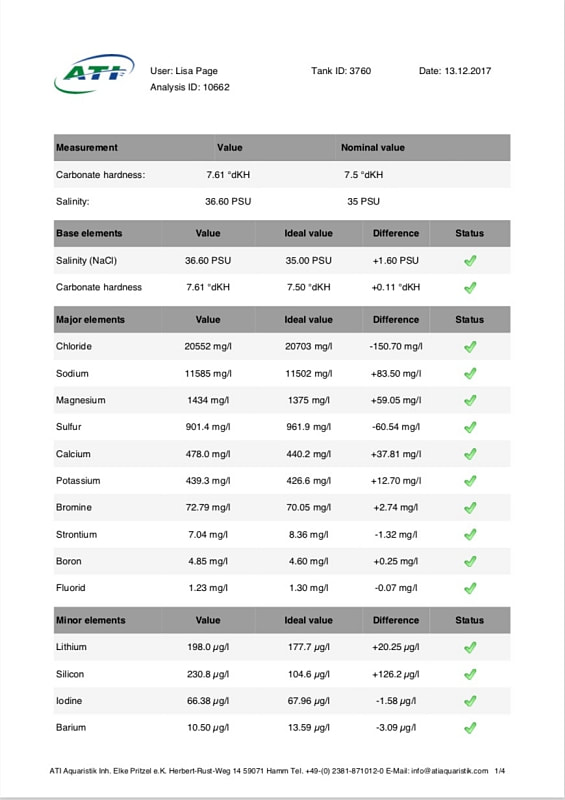
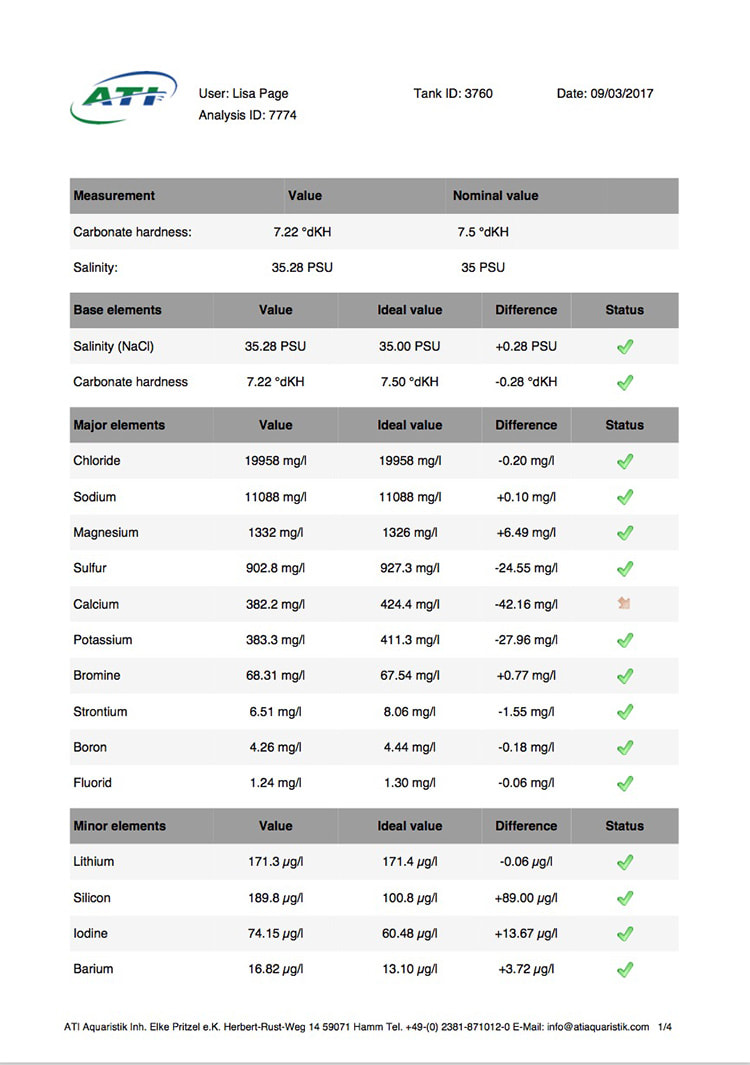
When the tank hit one year old I sent off for another ATI ICP-OES analysis. The results of which are below. Sooo, if it's not one thing it's another, last test the calcium level was a little low and now this time it's potassium. I don't currently own a potassium supplement and am debating whether I really need to order one now or not, I've never had to dose potassium before so it's a new one on me. The tin level is continuing to drop so that's one issue sorted and I seem to have hit on the right level of iodine dosing too. Nitrate and phosphate are still on the low side, I keep expecting them to rise as the tank gets older but there's not been much change yet. Despite this the coral growth is slow but steady and the colours are looking nice (well to my eyes they are at least) so I will continue as is for the moment.
Time for another ICP analysis to see how things stand with the Tin pollution as of 29th August 2017. I'm pretty happy with those results, just a tiny bit more tweaking required and everything will be right where I want it to be. The Tin has finally fallen to an acceptable level and hopefully will continue to fall until it's undetectable. The Calcium dosage needs increasing and the iodine dosage reduced a touch. One lesson learnt here is that the test kit used to monitor the iodine levels in between ICP tests (Salifert) is a complete waste of time. The iodine level in the tank has gone from 0 (mid April) to 74ug/l (end of August) and in all that time the test has registered nothing more than the faintest hint of colour which equates to less than 0.01ppm (10ug/l). I knew before I started that the home test kits for iodine were considered inaccurate but I didn't realise they were quite that useless (well the Salifert test anyway). I won't bother to use it again.
I'll sign off with an updated FTS (as of today) this time featuring a beautiful splash of yellow. My Halichoeres chrysus wrasse is pretty hard to miss don't you think. When I introduced her I had assumed that she was actually a he but I have been informed that these wrasses are all born female. So she now goes by the name of Rei instead of Ray and she will remain female for a while yet before making the changing to male on adulthood. |
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|





























































 RSS Feed
RSS Feed
