|
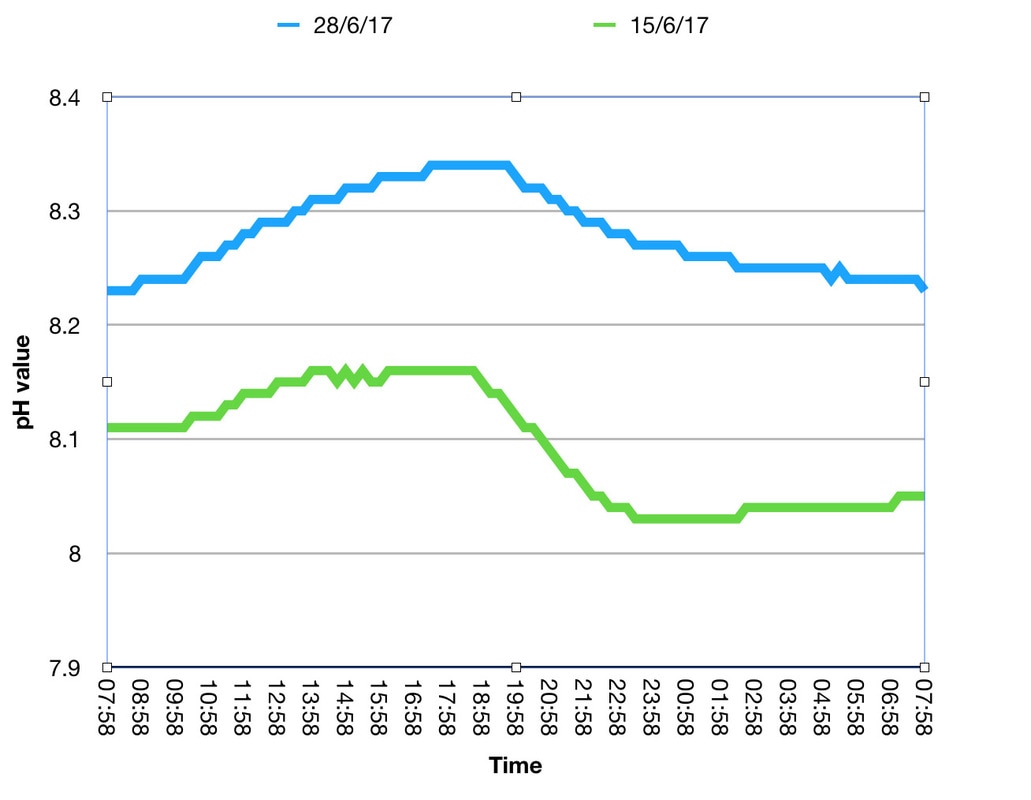
Since the addition of the Profilux controller I've been monitoring the pH cycle of the tank quite closely. I must admit that I just love this kind of thing, it's the scientist in me. I've noticed that the minimum value was lower when there were two of us working in the same room as the tank than when it was just me or even when there was no one in the room at all. When there were two of us working in there the pH would drop to below 8.0. I thus began thinking about adding a CO2 scrubber to my protein skimmer to see what difference it would make to the pH cycle. There are a couple of scrubber options on the market (that I know of) and after brief consideration I decided to try the new ATI Carbo EX filter since I have used their ICP test recently and the price seemed quite reasonable. Connection to my skimmer was easy and I have it situated in the cupboard alongside the tank (next to my RO resevoir). It measures just under 21cm high and 10cm diameter for anybody interested in the dimensions. As soon as the scrubber was connected I noticed that the tank pH began to rise, so it certainly appeared to be working. Frankly, I'm amazed that just breathing in the same room can have such an effect on the pH of a tank. I have drawn up a graph from two typical daily 24hr pH cycles, taken from 8am in the morning (when the lights switch on) to 8am the following morning. The green line shows data from the 15th of June (6 days before the fitting of the reactor, we were out of the house for half the day) and the blue line is from the 28th June (6 days after fitting the reactor, I was working in the same room for most of the day). As you can see the pH before the scrubber fluctuates from a low of 8.03 to a high of 8.16 and with the scubber it cycles between 8.23 and 8.34. Obviously this is data sampled from just 2 days but from from my general observation, the pH is most certainly higher with the scrubber and interestingly the cycle has a slightly narrower maximum and minimum range. Will this have a positive impact on the general health of the tank? That's the important thing, watch this space for updates!
0 Comments
I've just been having a quick play around with the on board flash on my camera in the hope of getting a reasonable shot of Crystal, my Bruun's cleaner shrimp (Urocaridella antonbruunii). She (or he, I have no idea) never stops swaying so taking a picture without flash almost always results in some motion blur. I expected the shots to be rubbish but was actually pleasantly surprised at the outcome. Next time I hope to catch her facing the camera instead of looking away.
Crystal is the best shrimp in the world! OK, I may be biased in that regard but she is a model citizen and has never bothered anything else in the tank. She doesn't try to steal food from the LPS corals or irritate them in any way. I know this can be a problem with certain other ornamental shrimp and I have certainly found that out myself in the past. Ugh, Lysmata amboinensis, never again! Crystal tends to be most active at night when she can be found swimming freely around the tank. During the day she likes to rest under her favourite rocky overhang waiting for the pumps to go off and signalling feeding time. She really has the most amazing red and white markings and how about those awesome 'wave form' looking spines along her back! I never knew they were there until I saw them in this photograph. Too cool! I have some new stuff. :o) After searching for ages I located a shop that had some Red Spotted gobies, Trimma rubromaculatus, in stock (finally Facebook is useful for something). They only had two left when I visited but apparently they were a mated pair so I just couldn't leave without them. They've been with me for 4 days now and seem to have settled in a treat. On introduction they were ignored by the other fishy residents with the exception of Candy, the Trimma cana goby. Candy is actually a male Red Striped goby and he was not best pleased to see a another male goby, even of a different species, invading his patch. There was much posturing between himself and what I would assume to be the male Red Spotted goby. No damage was done fortunately and now they appear to be keeping their distance from each other. The new gobies, named Rocket and Sparks, are much more active than Candy and are out and about a lot more especially at feeding times when the nanostream pumps are off. They do find it hard work to battle the flow when the pumps are on and are generally found suctioned onto the underside of rocks or resting on the back wall instead of swimming up in the water column. In addition to the gobies I also purchased another small frag of zoanthids, this variety is called "King Midas" and comes with some hitchhiking fan worms too, a nice bonus in my opinion. I think I'm pretty much out of space now where zoanthids are concerned. Picture to follow in my upcoming 1st July update. I also added to my crustacean collection in the form of a Pom Pom crab just because I think these guys are just the coolest. They have such beautiful markings and the little anemones they hold are neat. I hope he (or she) doesn't do too much damage waving them around the tank, lol! No photo as of yet because he's kind of shy at the moment. In other news Lurch the conch finally got up after his extended snooze. He spent almost 2 whole months hidden under the sand with no movement at all except for the odd glimpse of an eyeball and his proboscis poking out of the sand for an occasional bedtime snack. I'm surprised that he can survive for that long with such little food to sustain him. Luckily he seems none the worse for his 'hibernation' period thank goodness, I just wish I knew what caused it, is it a natural part of his lifecycle or was there some water quality issue that he didn't particularly like? The Nudus gobies have not been in much evidence since Gordon the Whitecap goby made his leap of faith last month. I used to see them all the time but after the upheaval with the pistol shrimp and the loss of Gordon they hardly ever came out of the burrow system and never both at the same time. Then Hop (the male) vanished entirely, the last sighting of him was on the 18th June and after that nothing. He has been known to go missing before, when guarding eggs, but I generally get to see his head pop out of the burrow every now and again. I was beginning to think that he'd had an altercation with the pistol shrimp and lost or been buried alive under the rocks, eek! Happily no, after 8 days he's back out again like nothing was ever wrong so I guess he had been guarding eggs again after all. I wish they'd let me know so that I don't worry so much, lol!
Just a quick snap of the Balanophyllia taken today with my iPhone after lights out. I've settled in to a routine of feeding this coral once a day now. It's not fussy what it eats, PE mysis, Krill, clam and mussels (which seems kind of wrong since there is a live mussel located right below it!) are all eagerly consumed.
The Montipora, despite my best efforts to kill these frags, have all survived and are showing signs of growth. :o) As for Acropora #2, there are no signs of any remaining red bugs (yet!), the remaining piece of encrusted coral looks like it may well survive to fight another day. The polyps are out at least, indestructible or what! I don't know what I will do if the bugs reappear, there are not many options left to me now other than just living with them or trying a pipefish for biological control. Acropora #3 continues to make a slow recovery from the blistering issue. The Plexaurella gorgonian has made an amazing recovery since it was moved to a different location. The damaged areas of branches have grown over already and it's pretty much back to its previous fluffy splendour.
I'd like to say that everything is on the up but just as one set of problems looks to be solved another rears its head. I've noticed that the Red Tuxedo zoas have not been expanding as they used to and some actually appear to be shrinking. Nudibranchs I wonder? Sigh, there's always something I find to worry about, lol! Anyway today I got my camera out and snapped some top down coral shots. I really want to get some new photos of the the fish, especially the wrasse but they are so very camera shy. Now that I'm down to four Red Spot Cardinals I've noticed that they no longer shoal together. Two of them hang together in the back left-hand corner of the tank, the third swims alone in the back right-hand corner and the fourth, the smallest one, swims right out at the front of the tank. I must admit I do miss seeing them all together. As for the Nudus gobies I've not got a clue what they are up to any more. They used to be out all day long but now they are mainly hidden underground doing goodness knows what. I wish I had X-ray vision so that I could see what they are doing. They are using the pistol shrimp's burrows; are they interacting with him at all? I would be nice to think that one day they might pair up but I can't see it happening at the moment Will, the blue-legged hermit crab shed his exoskeleton this week which reminded me that it'd been a while since I added any new shells. As soon as I dropped in some larger accommodation he marched up to the nearest one, whipped his naked bum out of his old shell and moved into the new one. Has he no shame, lol! Here he is showing off his bright white new shell soon to be covered in coralline no doubt. I noted in my June update that the Plexaurella sp. gorgonian had a damaged area on one of its branches. Well, a couple of days following that two more small damaged areas appeared and this time the gorgonin inside was exposed. The polyps then retracted completely. I waited for a week in the hope that it would miraculously recover but the polyps remained hidden and a layer of algae began to cover the branches. I decided then to take action and move the Plexaurella to a different area of the tank, if I left it where it was it would be a goner in no time at all. Finding a new location proved somewhat tricky as real estate in the tank is limited these days (ahem, what did I say about not over stocking my tank with corals!). In the end I decided to fix it close to where Acro #2 used to reside, it's not really an ideal spot long term, especially if the tiny remaining base of Acro #2 survives and sprouts new branches, but that's a problem for another day. Anyway I doubted that the gorgonian was going to make it but it appears I might be wrong. The following morning the polyps began to extend again and now 3 days later they are almost all back out again, it's not back to its previous fluffy glory but compared to how it looked before moving it's amazing. So was it a flow issue or a lighting issue? Or was the Seriatopora hystrix to blame, it was directly down flow of the Seri, maybe that was releasing some noxious substance that the gorgonian didn't like. Now I need to wait and see if the damaged areas can be recovered.
A coral update, first up Acropora sp. #3. In April I thought this coral was beyond hope, it was bleached and areas of the frag were dead. Then the lighting schedule was altered and things began to slowly improve. The lovely green colour returned but algae took a hold on the dead skeleton. Now, in June, the Acro is as green as ever and the flesh is regrowing over the bare skeleton. An amazing recovery which highlights just how resilient these corals can be given the correct conditions. Moving on to Acropora sp. #2, not such good news here I'm afraid. At the beginning of March I discovered that this coral was infested with red bugs, Tegastes acroporanus. I made the decision to try rid the coral of this pest by dipping it in Polyp Lab's Reef Primer, the only treatment I had in house at the time. The dip did work, killing the bugs and the Acro appeared to be pest free for weeks following with good PE. However, recently I've noticed that the coral had stopped growing and the polyps were absent again, Sure enough on close investigation the coral was covered with red bugs once more. Obviously some of the red bugs survived the first dip or remained in the tank jumping back on the coral after it was reintroduced. So, what to do now? In the intervening months the coral has encrusted at the base so removal was not a simple option. I can't deny that I wished that I'd just chucked the coral the first moment I discovered the little blighters thus (maybe) solving the problem straight away. After taking a big breath I deliberated the problem for a few days, revisiting the idea of dipping the Acro again and also the option of biological control. If I thought that adding a Dragonfaced pipefish was a sure fire fix and my tank could have supported one I would have been setting up some copepod cultures straight away in preparation and given one or more a try but it's such hit and miss affair. Some individuals eat them and some ignore them and I cannot in all conscience add a fish that might not thrive just to fix a problem of my own making. I should have QT'd the coral or least dipped it before adding, sigh! Never trust an LFS for a healthy pest free frag! In the end I opted for removing the coral as much as I could before a water change and scrubbing the remaining encrusted base whilst siphoning the water at the same time. Either the encrusted base survives without the bugs which is a perfect result or the encrusted base dies but I'm red bug free which is acceptable or worst case scenario the red bugs are present on another coral in which case I have destroyed a perfectly viable frag for nothing. Stay tuned for updates if you dare.
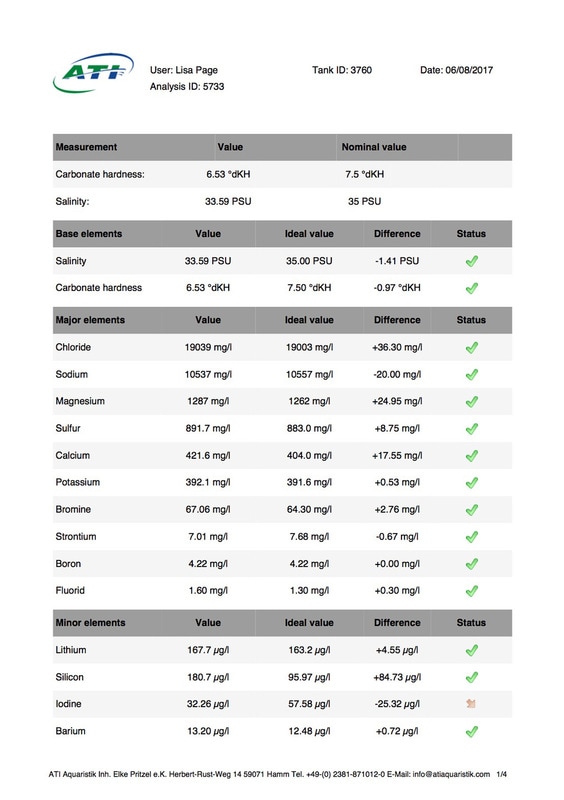
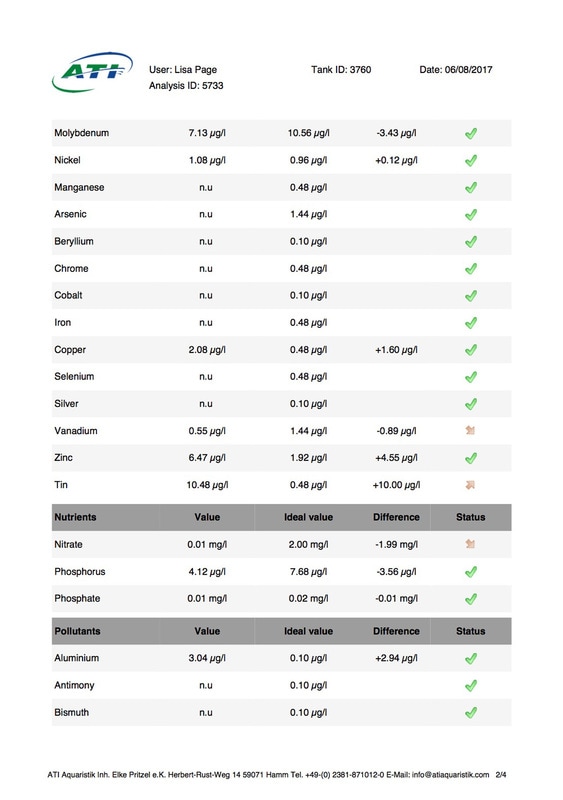
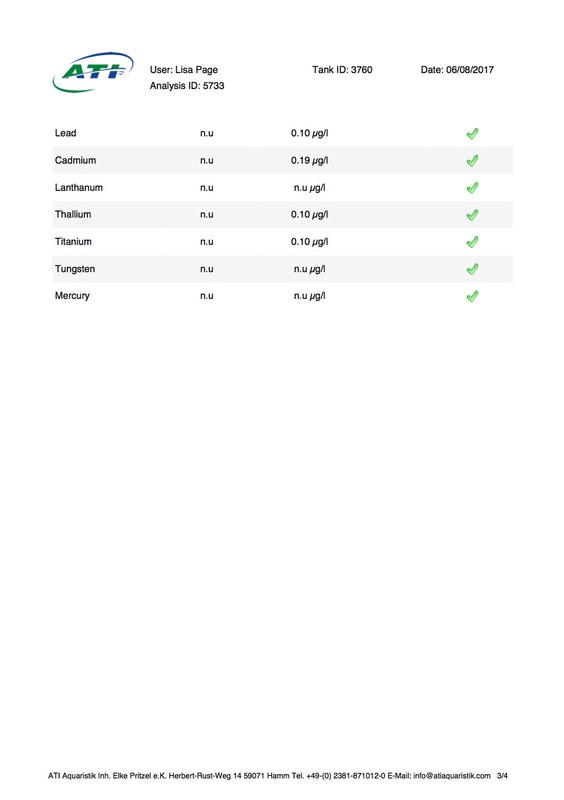
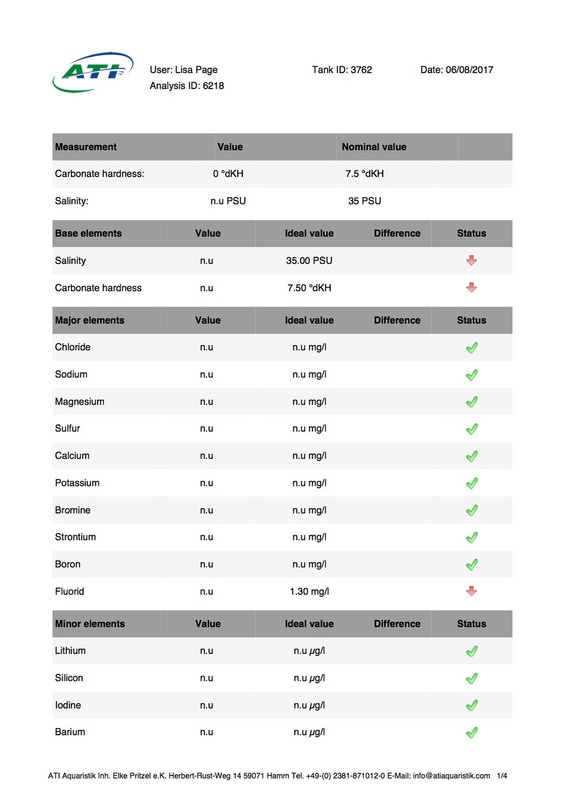
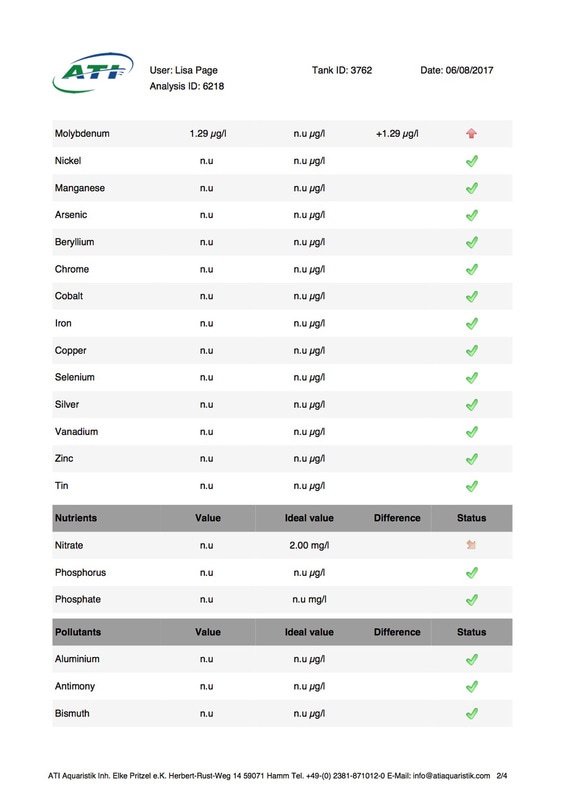
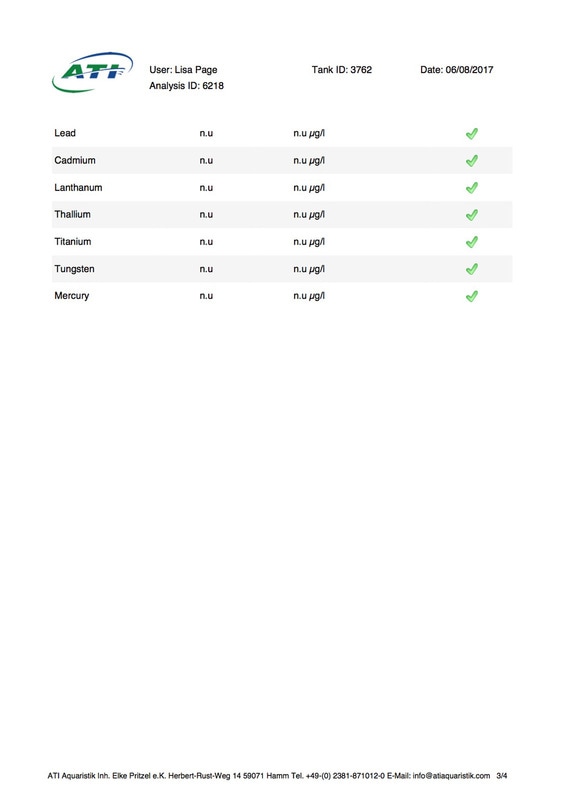
It's been 6 weeks since I sent a sample of my tank water off to Triton for ICP analysis. My maintenance routine has been pretty much the same since although I've been changing out slightly more water than usual (12-14% as opposed to 10%, I would perform more if I could but the buckets I use only hold 25l so I can't perform larger water changes easily), I've also been dosing trace elements, in particular iodine as it was undetectable. I thought it was time to see how things stood now. I was going to use the Triton test again for continuity but discovered that ATI offer a test for RO water in addition to tank water analysis so it was a no brainer for me, ATI was the obvious choice. I have always wanted to know for sure that the RO water I use is without contaminants. First up is the tank water analysis: As you can see I still have an issue with tin, if the Triton and ATI ICP machines are compatible then you could say it's dropped a little (from 12.00 to 10.48ug/l) but as far as I'm concerned there is not much change at all. I do have an iodine reading now which is good news it's still low but a lot better than 0 (at least I'm assuming it is, lol). My nitrate and phosphate levels are still low despite feeding, what seems to me, a lot of food. The Siporax and/or refugium appear to be working well, a bit too well it would seem. Now on to the RO analysis, results below: I am happy to see that apart from a touch of molybdenum the water looks squeaky clean, not a whiff of tin which is reassuring. Water changes must be helping keep the tin level from rising further but not enough to actually lower the level, I must assume that whatever is leaching the tin that it must still be doing so. Almost all of my equipment is brand new with the exception of a couple of small pumps running the refugium and the pump that I use for water changes. They look fine but I've decided to change them out just in case. I replaced the old glass heater that I used in my water change bucket last month ago as it seemed like an obvious suspect.
As far as the corals go they seem to be doing OK at the moment with no sign of any flesh stripping which apparently can result after long term exposure to a high level of tin. I hope I can find the source and fix it before I reach that point, assuming of course it's not coming from the glass itself in which case I'm stuffed. I went away this weekend and managed to find the time to visit a few not so local fish shops (as the reef-obsessed tend to do given the opportunity). In the 3rd shop I struck gold and located the Porcelain crab (Petrolisthes galathinus) I've been searching for since I first set the tank up. She is one of the hitchhiking species that are sometimes found in live rock. I used to have one of these peaceful filter feeding crabs in my old tank and knew I wanted one for this tank too. When I arrived back home everything in the tank seemed fine and my crab (a female according to the shape of her abdomen) was duly acclimated and introduced just before the lights went out.
This morning I eagerly rushed down to check on my new crab only to discover that the curse of the Red Spot Cardinalfish had struck again. Another one of them had died, this time I discovered the body, the Lobophyllia was trying its best to eat it! The meal turned to be too much for the small coral to stomach and I was able to remove the dead fish using a pair of tongs. It was at this time that I happened to notice a small shape lying on the carpet to the side of the tank. Closer inspection showed it to be Gordon my Whitecap goby, nooooooo! He had jumped out of the tank at sometime during the night. I do have a mesh lid on the top to prevent any jumpers but somehow he still managed to find a way out, he was a very small fish after all. There is no doubt in my mind that he jumped as a direct result of the persecution by the Nudus gobies. The female in particular went out of her way to terrorise him at every opportunity. I have no idea what will happen to Al the pistol shrimp now and I dare not introduce another Whitecap as the same thing could happen all over again. He's going to have to make do on his own from now on and I expect I won't see him nearly as much as when Gordon acted as lookout for him. I feel so very, very sad today. Another month has passed by so it's time for another update. The day after I added the three gorgonians etc. I received one more delivery. This time the box contained some tiny frags of encrusting Montipora danae on frag plugs plus a small rock of fan worms. When it came to mounting the Montipora frags I removed the base of the plug but that still left huge round chunky base, no matter how it was positioned it would never, ever blend in. So I decided to take the rather risky option of removing the frag from the plug entirely and sticking it directly on to the rocks. Since the frags were only 1 cm across it's not hard to imagine how beaten up they were during the whole process, I don't think that I've ever treated corals so harshly in my life, well not intentionally anyway. This is what they looked like 2 days after fixing down..... ...and here's how they looked 9 days later, amazingly not dead yet. The green one looks like it will do OK the other two not so much, only time will tell if they make it or not. For the most part the other corals have been doing fine with base encrustation and/or upward growth. The zoanthids are really doing great with multiple new head; the exceptions being the first addition which seems to stay the same no matter what and the Wango Tangos which still have the same number of heads as when they went in with. The clam seems happy with its lot and has developed an extra line of scutes since I've had it. The only coral I'm specifically concerned with at the moment is the Plexaurella gorgonian. I noticed that it had a damaged area on one of the branches yesterday, I'm not sure if it's because it's unhealthy or because I've inadvertently damaged it. I do get very close to it with the pipette when I'm feeding the pistol shrimp so I will take more care in the future and hope that it can recover. There is a bumper photo update below and I've added the new shots to the individual livestock pages to illustrate any growth (or not, lol). There are a few shots left to take such as the clam but that will have to wait till another day. I'm pleased to report that I haven't lost any more of the Red Spot Cardinalfish which is a relief. Sadly there has not been any more spawnings, I see plenty of what looks like courtship behaviour but no release of any eggs.
The Nudus gobies have unfortunately been evicted from their cave yet again by the pistol shrimp's digging activities this has been upsetting for everyone concerned. I really need to remove either the shrimp + goby or the Nudus goby pair but I'd never be able to catch them without stripping out the tank which I just can't bring myself to do. I've hardly seen or been able to feed any of the affected parties for the last 2 weeks. Skip the female Nudus goby vanished for the 3 days, leaving Hop, her mate, swimming about searching for her. I feared that she'd come to a sickly end facing up to the pistol shrimp but to my very great relief she popped up again ready to fight another day. She'd probably spent the 3 days underground hassling the Whitecap goby who always seems to be the biggest loser during these turf wars. I hope he's survived OK. I can still hear the pistol's shot every now and again so I know he's fine. I'll sign off now with the obligatory new month FTS. I took it upon myself last month to strip and clean one of the Tunze nanostream pumps. Boy did it look out of place when it was put back in, I suppose I should do the other one to match but now that the coralline has gone it's allowed the brown algae to grow. The snails need to get munching pronto. |
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|


























































 RSS Feed
RSS Feed
