|
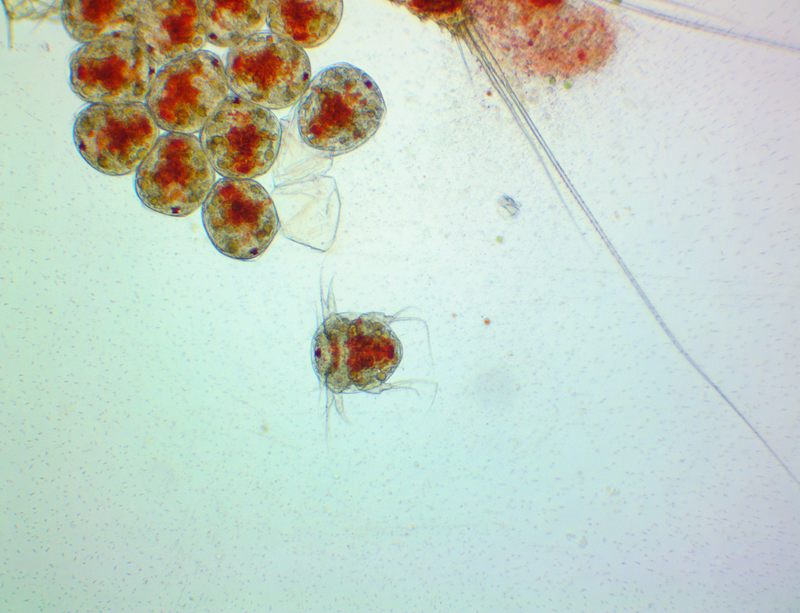
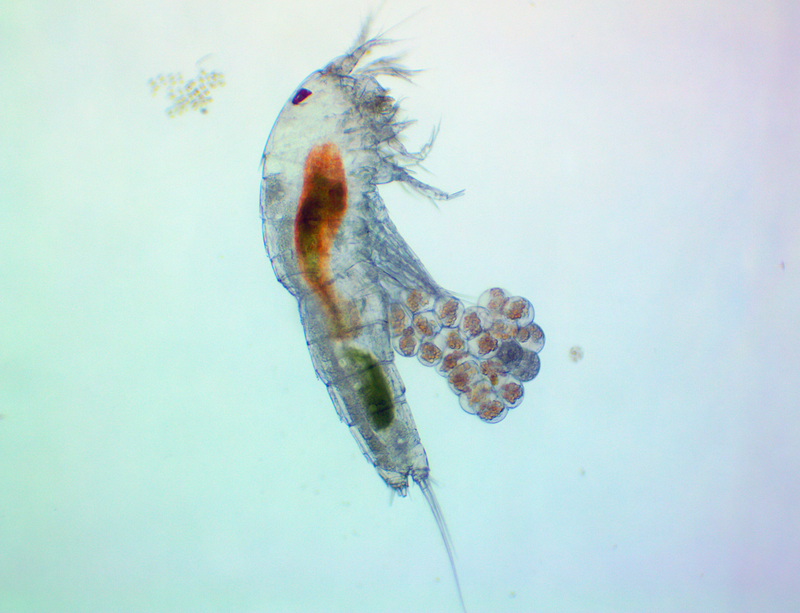
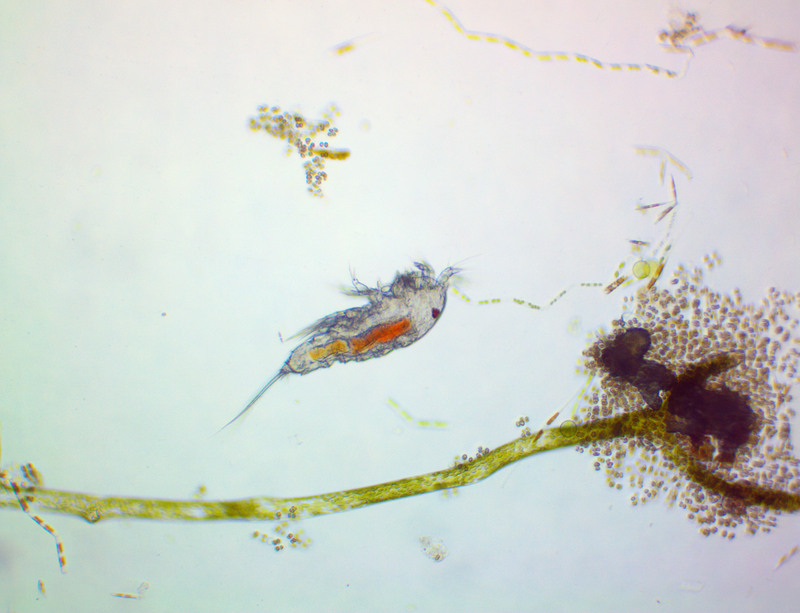
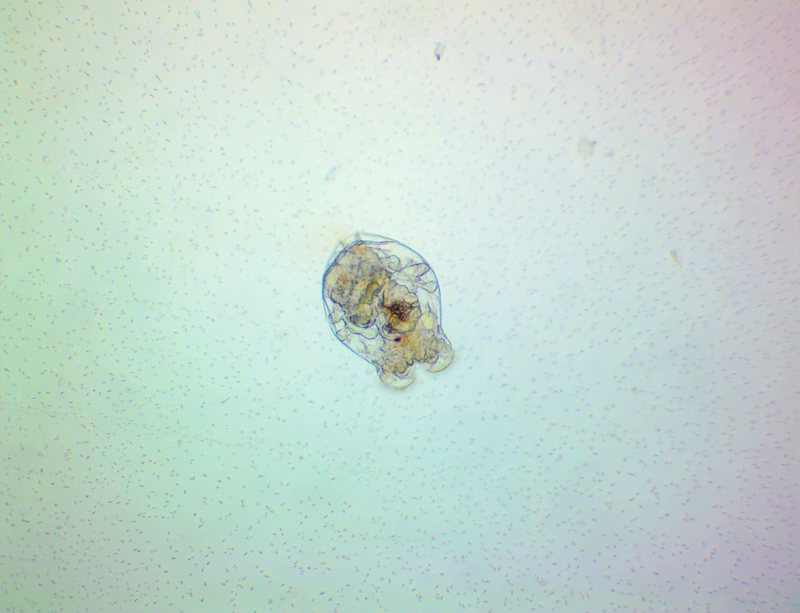
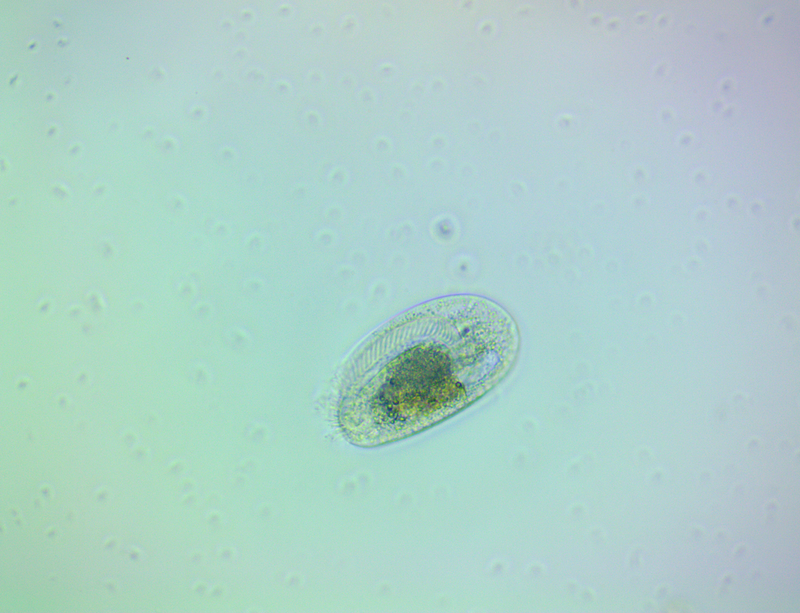
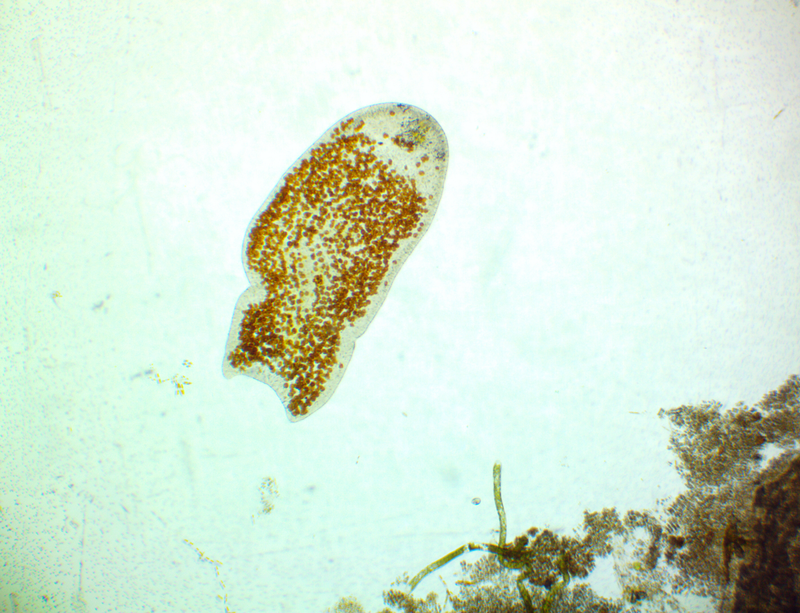
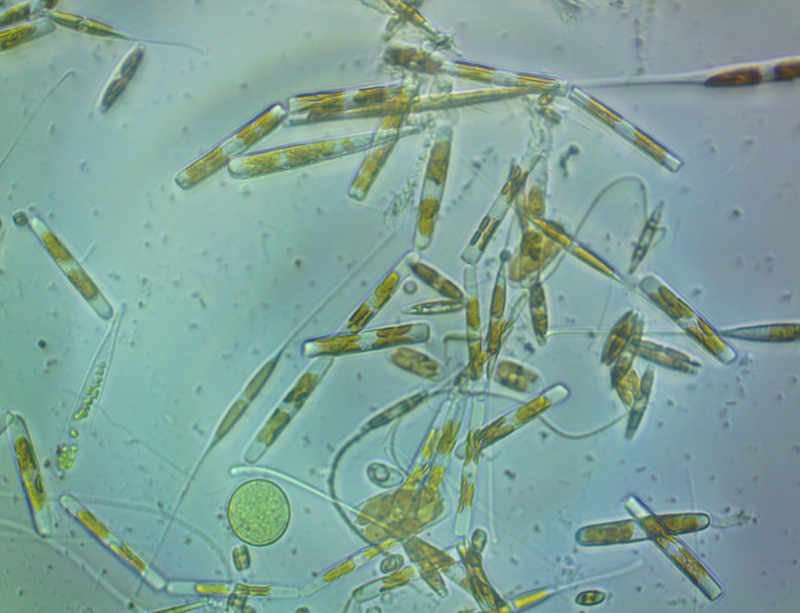
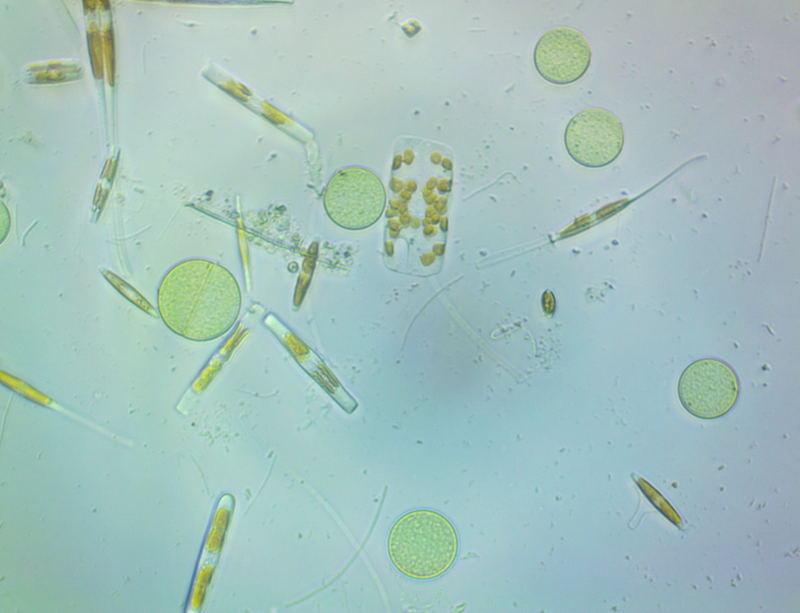
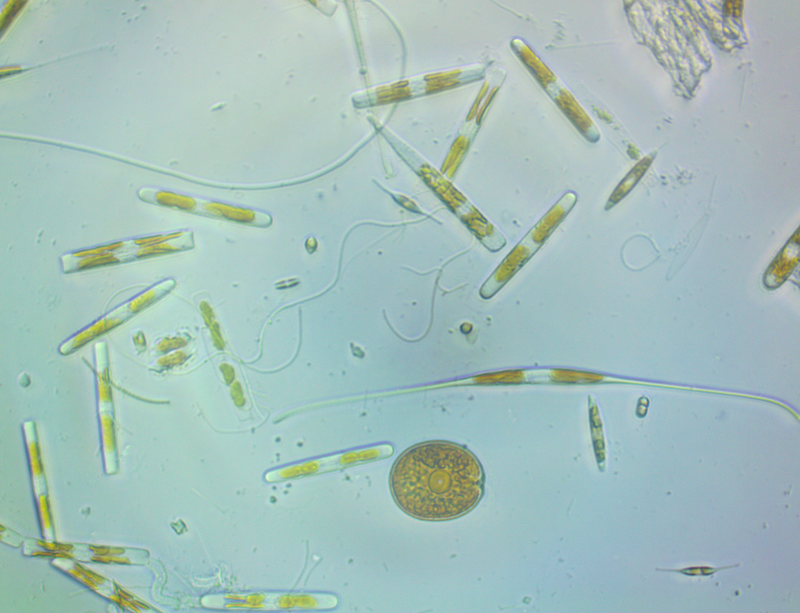
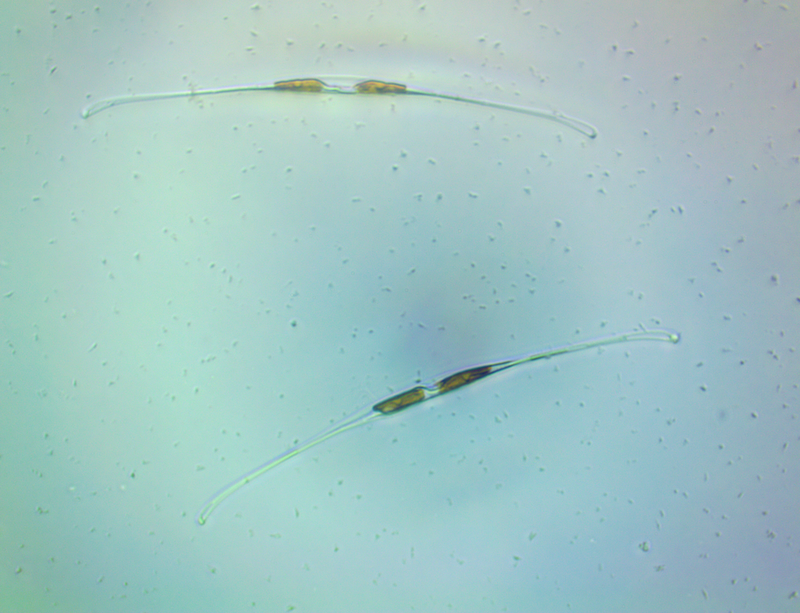
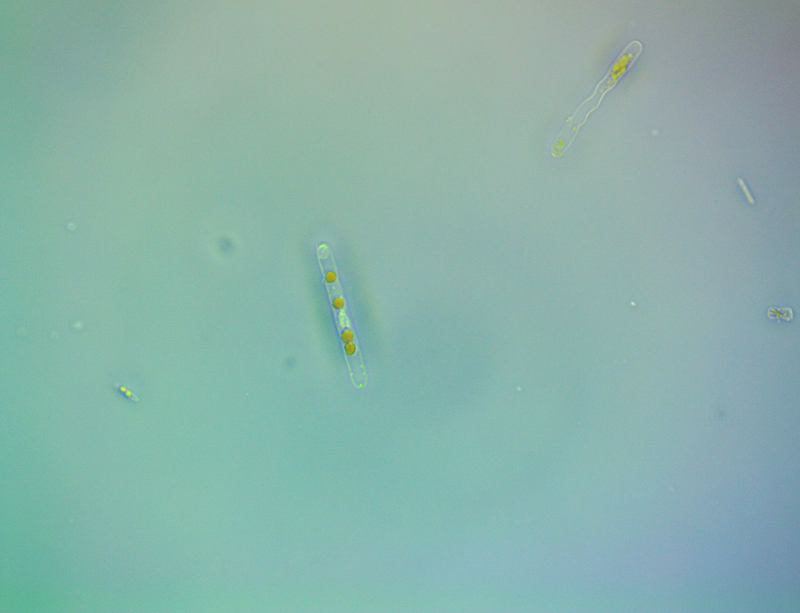
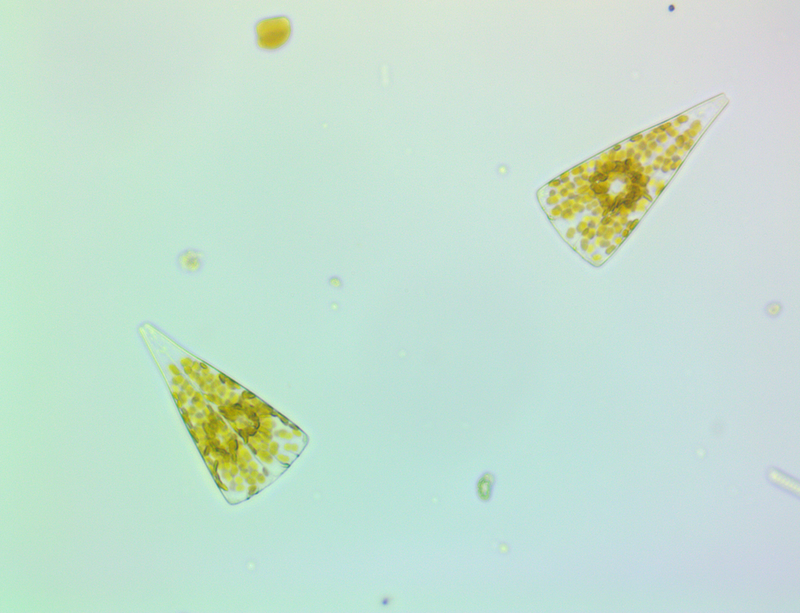
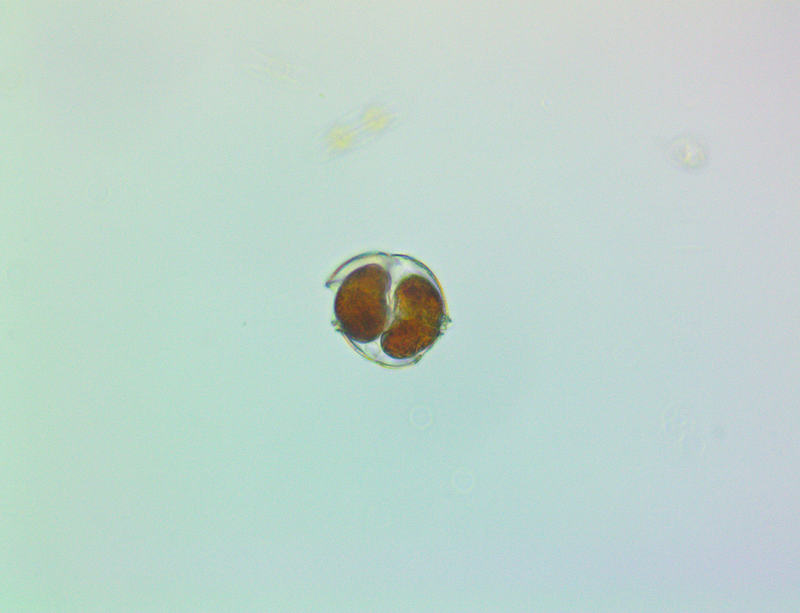
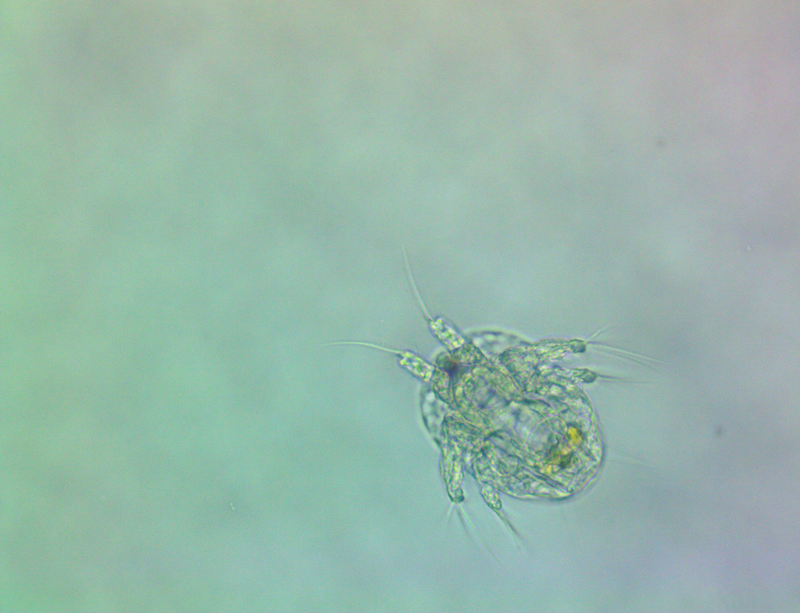
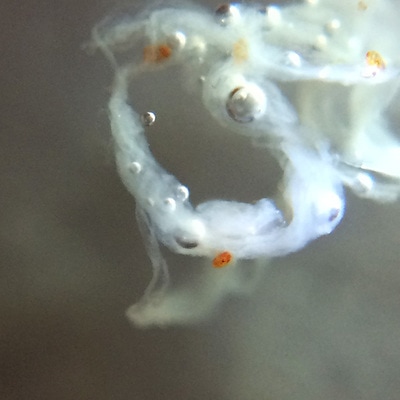
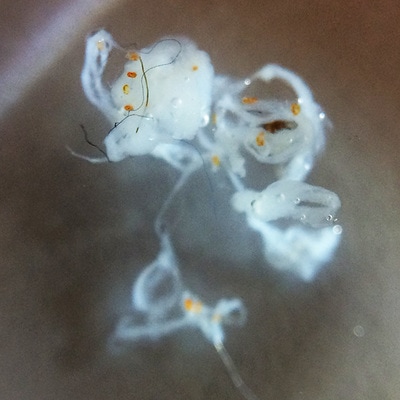
It's amazing the life that can be found in just a drop a water. Here are just a few microscope shots of the other fascinating (to me at least) microorganisms that I have discovered whilst on my dinoflagellate eradication mission.
0 Comments
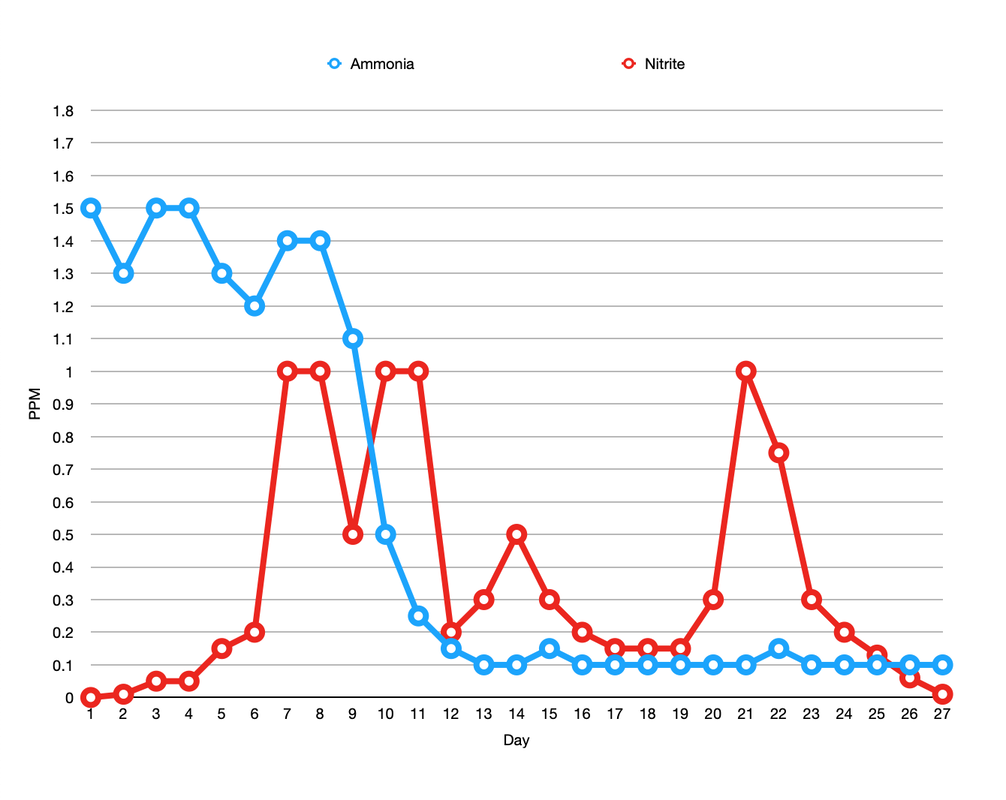
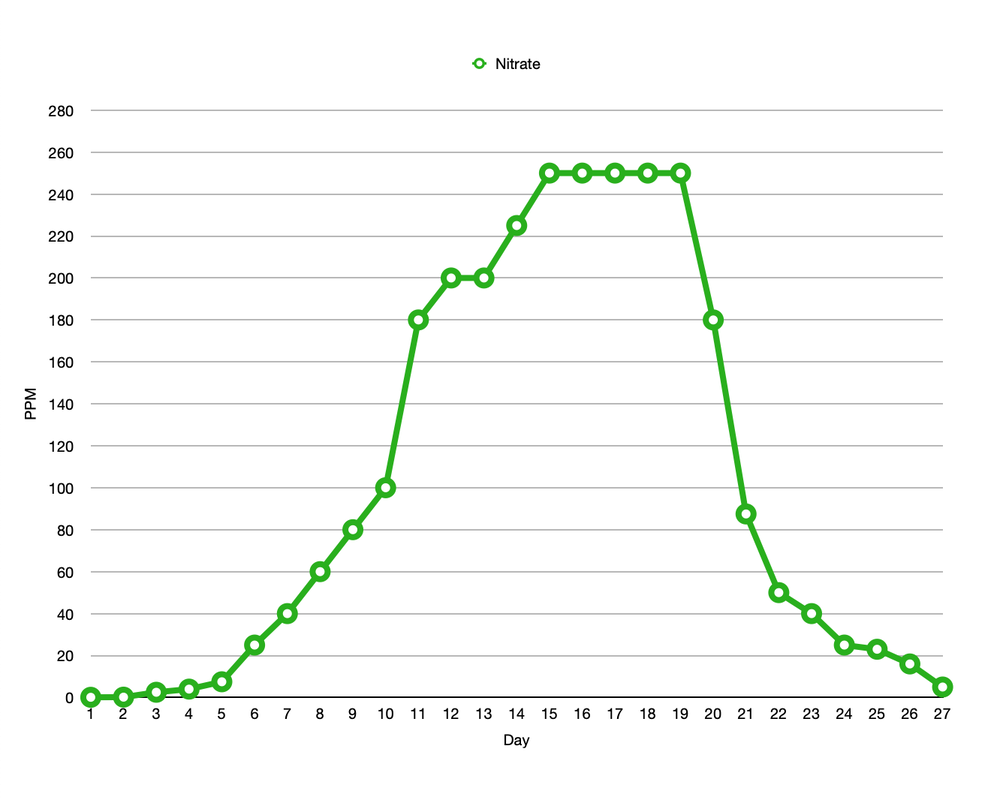
I began the tank cycle on the 2nd February 2020 with a bottle of Dr Tim's One and Only plus the recommended dose of ammonium chloride. Thereafter I tested ammonia, nitrite and nitrate every day for 27 days. Needless to say the levels I recorded in my tank did not look like the neat and tidy graphs I see posted online. I added further doses of ammonium chloride, as per instructions, on days 3 and 6. I also boosted the ammonia again on day 15 and 17 to keep things ticking over. The lights were left off during the cycle but there was some overspill from the Reefer 170. Here's what the ammonia and nitrite levels looked like. And here is nitrate. By day 10 the nitrate level had reached the upper limit of my Salifert test kit so I had to dilute the sample first by 1 in 5 and then later on by 1 in 10. On day 15 the nitrate level had reached a whopping 250ppm!! I envisioned having to perform water changes every week for the next 6 months or so. Things were starting to look a bit grim. The largest water change I can perform at any one time is 30% so on day 19 that's exactly what I did. The following day I recorded the level at 180ppm which is exactly the amount I expected following the water change but what I didn't expect was to see an increase in the nitrite level again. I continued my daily testing and not only did the nitrite levels fall back down again but the nitrate levels kept dropping and dropping till on day 27 they read a rather acceptable 5ppm with no further water changes required. How weird! I have a feeling that the off the scale nitrate levels were probably due to interference by nitrite, either that or I have a super duper nitrate reducing system in my tank. Patent pending. Whilst the tank was cycling my tank cover arrived and I was somewhat disappointed to discover that there is what I consider to be an excessively large gap between top of the weir comb and the mesh lid. What's that all about? Why can't the level of the weir be designed to be more in line with the top of the tank?? I don't have the D-D branded cover but I can't imagine that sits any lower than any of the other alternatives. Anyway there's more than enough room for any of my small to moderately sized fish to squeeze through and since there is no cover on the weir they'll end up stuck down inside it or take a ride down into the sump. Neither alternative is acceptable so I plugged the gap with some clear acrylic sheet. There is still a small gap to the right of the weir comb that I haven't fixed but it's pretty narrow so hopefully my fish won't discover it (famous last words I know). On day 11 I fired up the skimmer, an Ultra-Reef UKS-160 mainly for aeration purposes. This is not a new skimmer but you wouldn't know it, as soon as the pump was fired up it went completely crazy so I left the cup off for the time being. Plenty of air going in there to be sure. 10 days later it showed no signs of calming down and I was getting a bit sick of the salt creep everywhere so I fitted the cup with the drain plug removed so that the water overflowed into the cup and back into the sump again. As you can see protein scum began collecting inside the cup. Skip forward another 13 days and still the skimmer was in overdrive despite the flow restrictor being wide open and the unit sitting in exactly the right level of water. I decide to craft a stand to raise it up a bit, eggcrate, PVC pipe and cable ties later...ta da! That was version 1, since then the pipe legs have been cut down a bit as the skimmer was then sitting a bit too high (typical) but it's started working well now. I have read that skimmer collection is reduced in tanks where roller filters are also employed but it's definitely pulling out some stinky stuff. Talking of the X-filter, I must hold my hand up and admit how I eagerly watched and waited for the motor to turn for the very first time, yes I'm sad like that. It seemed to take an age for the water level to rise up and trigger the level sensor, lol. The excitement has since worn off a bit as I've become used to the motor just doing its thing, it is quite satisfying to see the used dirty brown roll. Better out than in (I hope). On day 27 I considered the tank to be pretty much cycled. Diatoms were beginning to take hold on the sand and rocks so I decided to add some Tisbe copepods. I wanted to encourage the growth of these little critters as much as possible before transferring any of (my pod loving) fish over from the Reefer. For a couple of days I thought the tank must be toxic and I'd killed them all off as I couldn't find a single pod on the glass but then they bit by bit they slowly started to appear. This made me one extremely happy reefer (I'm easily pleased clearly). On the same day as adding the copepods I fired up the refugium. I'm a big fan of refugia not just as an area for the growth of macro algae but also for the reproduction of beneficial critters (and also as a place to relegate any naughty crabs etc if needed). I wanted to keep the light spill down to a minimum inside the sump itself so I designed something to sit in the cupboard to the left of the sump with the pipework entering and exiting via the existing hole in the cabinet. I didn't want to have to modify the cabinet in any way and I almost, just almost, got away with it. The pipework was a tight fit but worked out perfectly, however the tank height was out by just over a millimetre, arghh! I had to sand down the back wall of the cabinet a touch to get it to fit. Ooops! I decided not to make the tank myself this time as it would be a lot bigger than my previous set up and hence more water to worry about leaking, instead I got Wharf Aquatics to make it for me. I'm running a Kessil H80 for the time being (that one I had lighting the Reefer refugium) but I may switch to something more viewer friendly at a later date, I'm not really a fan of the red/purple lighting to be honest. Here it is looking clean and tidy (needless to say it doesn't look like this now, far from it!). Incidentally even though I did not directly add any copepods to the refugium some have made it down there anyway from the DT and it's now buzzing with pod activity. I have the X-filter bypass controllers fully closed but still the pods have managed to find a way through the filter, past the skimmer and through the refugium pump/pipe and into the refugium, which I find pretty amazing to be honest.
I have always had a hankering to try a non-photosynthetic gorgonian, it's crazy I know because feeding them can be so hard but I think they are incredibly beautiful corals. Apparently out of all the species available Menella sp. is reportedly the easiest to keep however in all my years of running a reef tank I have never seen one for sale in a shop. Then amazingly a tiny frag became available online. I reasoned to myself that surely I could find somewhere to squeeze in something that small and before common sense could prevail an order was placed. The frag arrived looking great and the polyps were fully extended in the pot, I placed it on a frag rack whilst I pondered where on earth I was going to put it. It really is lovely, delicate yellow polyps extending from a dark red central stalk. I have been offering it a variety of dried and frozen foods not really knowing what it prefers to eat or is indeed the correct size for it to swallow. Out of curiosity I decided to film how it would react when I dumped in a load of live Tigriopus copepods in to the tank. I didn't really expect to see much so I was quite surprised to actually catch some action when played back. The footage is not great having been digitally zoomed in on my phone and then cropped on the computer plus the speed has been slowed down but you can actually see pods being caught and more excitingly being ingested! Keep an eye out for the pod that gets caught on the right hand side of the branch and at the time stamp 1.02 you can see the polyp actually swallowing it! Pretty cool stuff I think. Time will tell if I can manage to keep this beauty alive long term. Now that it's fixed in place I should be able to track any changes be they good or bad.
Soooo, I've been keeping an even closer eye on the tank (if that's possible for me, lol!) ever since I noticed one of the Acropora corals (#3) had blisters on it's branches.
Acropora #2 had been showing less polyp extension than the others but it's been growing fine and the colour has been good with lovely blue tips to the branches. I thought it was doing fine, however whilst processing my last set of photographs I happened to notice some red dots on the branches of the coral. Uh-oh! Alarm bells instantly started ringing in my head. Out came the magnifying glass and lo and behold there they were, red bugs!! Honestly this tank must be in the running for the award for the introduction of the most number of undesirable hitchhikers in the shortest amount of time. I have not had the pleasure of having to deal with these parasitic crustaceans before but I do know about them. Red bugs (Tegastes acroporanus) are tiny predatory copepods that live specifically on Acroporids. I considered my options: 1. Do nothing, the coral looks fine and is growing even though PE is almost non-existent. 2. Treat the tank with the recommended medication, Milbemycin Oxime (known as Interceptor or more recently Sentinel in the US). It is a prescription only medication designed to treat dogs suffering from heart worm disease but also happens to kill marine crustaceans. 3. Remove the infested coral to a separate tank and treat with the above. 4. Remove the infested coral to a separate tank and treat with a proprietary coral dip. 5. Take the coral out and throw it in the bin. I wasn't a fan of option 1, the coral may be fine now but it's never going to thrive whilst covered with parasites. Option 2. was entirely out of the question, Milbemycin kills ALL crustaceans so my crabs and shrimp would also be toast. I could remove my precious crabs and shrimp to another tank but to catch the pistol would require removing most of the rock work. That means major stress for the tank and major stress for me too! I love my shrimp and goby and there is no way that I'm going to risk upsetting them (and the rest of the tank) unless absolutely necessary. Option 5 was seriously tempting, it's only a small frag at the moment so it'd be no great loss but I do hate to give up without a fight. Plus there is really no way to know if any of the other corals are parasitised too, trashing one coral may turn out to be pointless if others are also affected. So really that left options 3 and 4, option 3 being preferred as Milbemycin is known to definitely kill red bugs whereas a coral dip may not. So with the decision made I just had to source Interceptor or whatever it is called in the UK. Unfortunately that plan soon went out of the window as our regular veterinarian refused to prescribe it for me, so it was back to the drawing board. The only coral dip that I had to hand was Polyp Lab's Reef Primer and after much scouring of the web I couldn't find any mention if it was actually effective against red bugs or not. How typical! Still it's all I had to hand and I thought there was nothing to lose by giving it a try. I mixed a little over the recommended dose of the Primer in a container of tank water, removed the coral from the rock work and immersed it carefully. Almost immediately the Acro started to exude excess mucus. I continually squirted the Primer solution over the branches with a pipette for about a minute and then checked the coral with my magnifying glass, I was surprised to see red bugs trapped within the mucus and no longer moving. Encouraged I removed all the slime (with bugs) that I could see and carried on blasting the coral with Primer solution. After approximately 5 minutes I moved the coral to another container of tank water, rinsed it off and placed it back in the tank. Unsurprisingly the coral looked rather pale by then. Today Acropora #2 looks pretty darn good, it's still a little pale and has no PE but it's alive and more importantly there are no red bugs on it! I've checked multiple times throughout the day and cannot find a single one. Of course that's not to say that my tank is now a red bug free zone but it's a step in the right direction. The other Acros seem unaffected and have good PE so you never know I may actually be lucky. Heh, considering how things have been progressing in the pest department probably not, but still I do like to be optimistic. Hmm, I did have to remove 2 more tiny Aiptasia from the sand yesterday, discovered in the same spot as the one I found back in January...... Finally I can confirm that the Whitecap goby and pistol shrimp partner are still alive. Yesterday the hole at the front of the tank reopened a tiny amount and an antenna popped out and waved around, additionally there were several shots heard throughout the day. Phew! There was no sign of the goby however as the hole was too small and awkward to see in to. Can a tiny fish like that survive sealed up underground for 6 days without food? I honestly thought not, I am so pleased to say I was wrong about that. I guess there could be small critters such as copepods to snack on but it's hard to imagine being confined in the dark with no route in or out for days on end, fine for the shrimp perhaps but what about the goby??
Anyway today at the first feed of the day the front hole opened up a little again, at the second feed it got a bit wider and lo and behold a white head appeared below. Whoop whoop! I got to see the shrimp and fish at the third AND last feed of the day too. Three sightings in one day, crikey I'm not sure I can cope, lol! Now I just need to see the whole body of the fish to know if it's skinny or not and to take a photograph of the pair together of course. I can dream can't !! Needless to say the Whitecap has now been named Gordon (from the film Flash Gordon, gotta love Brian Blessed's catchphrase). :o) My other good news is that Kylie the Pink Streaked wrasse has also begun to feed and is now starting to hang out with her new best buddy Edna the Possum wrasse. She still looks a bit unsettled but we are heading in the right direction so all is good hopefully. I need to clean the glass now ready for a new FTS tomorrow, wow January passed by really fast. As expected, the hole had not reappeared at the front when I got up on Saturday morning, however some other serious sand moving had taken place overnight. I guess the T. nudus gobies will not be moving back into their cave anytime soon because it no longer exists, it’s been totally filled in with sand. I now refer to it as the wall and not the cave. I may not be able to actually see the pistol shrimp much but it’s certainly making it’s presence known within the tank.
When it came to the first feed of the day to my very great surprise as soon as food hit the water a crater appeared in the sand just in front of the wall and a claw popped out, how very, very convenient! I was able to drop a piece of mysis right into the hole whereupon it quickly vanished. I dropped another piece in hoping to catch a quick glimpse of the Whitecap but sadly I did not. The view is not the best as you have to peer at an angle down through the glass again and the burrow vanishes under the rocks but still I’ll take that. I hope he stays put and works on it some more. I am hoping to be treated to views of the goby hovering at the lip of the burrow with the pistol in constant contact with his antennae hard at work shovelling sand. Ha, we’ll see! Sadly at the moment the hole does not remain open very long, the movement of the conch and other members of the clean up crew fill it in very quickly. I managed to get brief glimpses of the shrimp at later feeding times but nothing of the Whitecap. This morning the same thing happened at feeding time, the ‘sinkhole’ reopened and a claw appeared at the bottom, but where is the fish?! I need to see the fish too just for my peace of mind, I hope he’s still OK in there. Right, on to other reefing matters. I decided to whip out the algae magnet this morning and give the glass a much needed clean, I’ve only done it once since the pistol and goby pair were introduced in the hopes of allowing them time to settle. Anyway a layer of algae had built up allowing the copepods to flourish. There were literally hundreds of them! I actually felt bad squishing them with the algae magnet. Edna clearly needs some help eating them. Anyway it was at that time when I was taking extra care not to pick up any grains of sand and scratch the glass when I something new caught my eye. An Aiptasia!! Sitting there brazenly ‘growing’ out the the sand bed. Where on earth had that suddenly come from?! OK, I’ve only added two sets of corals, the first coral came with zoanthid munching nudibranchs, now the second with Aiptasia. I can’t wait to see what’s going to arrive with the next lot, planaria or a Eunice worm perhaps?? Now that's something to look forward to, rofl! Sadly there's still nothing much to report on the new fish and shrimp additions. I've not seen the Whitecap since Monday night when it came out and swam around after the lights went out. I did add another dose of live copepods last night in the hope that they might tempt the fish out to eat, but no joy. I can't deny that I am fearing the worst for the little fish now. With regards to the shrimp I did catch a tantalising glimpse of an antenna poking out of a tiny hole at the base of a rock on Tuesday morning, it was a blink and you could have missed it moment. Gosh, what I wouldn't give for x-ray vision right then (and indeed now!!). Who knows what's going on underneath those rocks? The shrimp has been oddly silent since the first night and there's not much in the way of observable digging activities either. Having said that a hole has re-appeared in the sand at the back of the right-hand rock pile today which I can only assume is the pistol shrimp's handiwork. Unfortunately there was a T. nudus goby sitting at the top of the hole and not the much longed for Whitecap, argh!
Edna the possum wrasse is settling in well, still a little shy but out and about much more than I expected her to be after only two days. She is tucking into brineshrimp and mysis as well as helping herself to the copepod buffet available in the tank. I anticipate the numbers of pods will fall dramatically now.
|
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|






















 RSS Feed
RSS Feed
