|
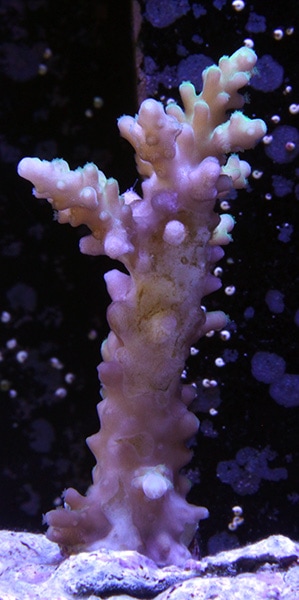
An interesting month to be sure. Generally the tank has been fine with no losses but there's just been something not quite right with some of the corals. The colours had been fading and polyp extension was poor. On the 14h April I decided take action and alter the lighting profile/ intensity to see if it would help the corals. In no time at all the corals started to pick up, the colours looked a bit better and the polyps began to extend again. It's only been 17 days and there is still a way to go but the corals do look happier and healthier. The worst affected was Acropora #3, not only had it been losing colour but it was also suffering from blistered skin. The photos below show how the coral has changed over time: Fingers crossed it will make a full recovery assuming I can keep the water quality up to scratch. Now for a few more coral shots: Last but not least the obligatory FTS. Unfortunately I was so intent on trying to get all the fish in shot at the same time (except the Whitecap goby, there's no way in a million years that would be in the shot, period!) that I didn't make sure that they shutter speed was fast enough to prevent motion blur. Ooops!
0 Comments
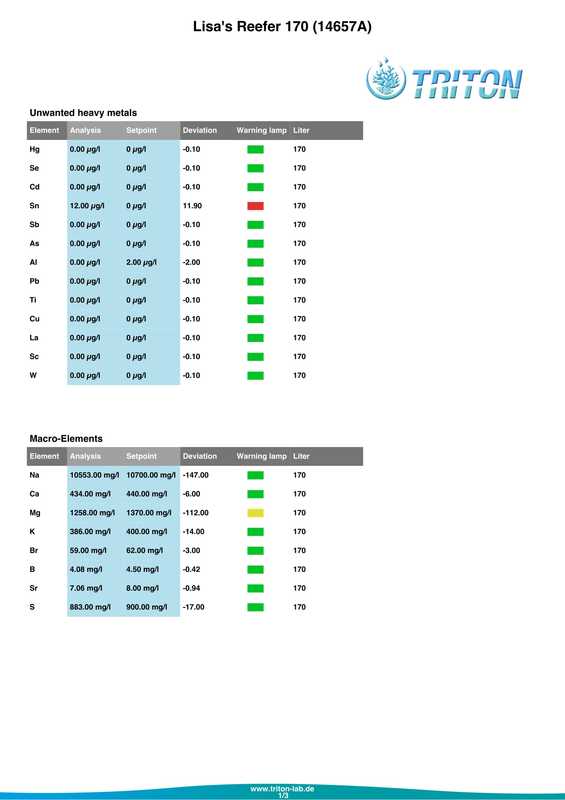
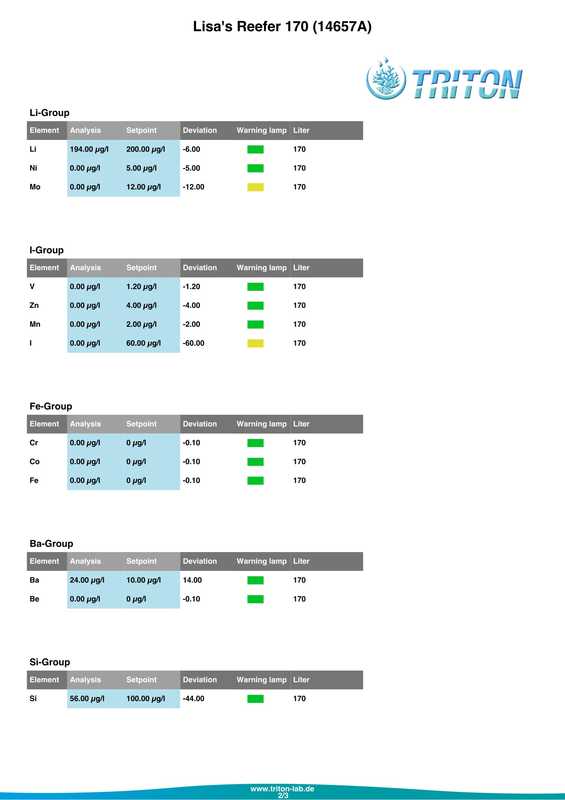
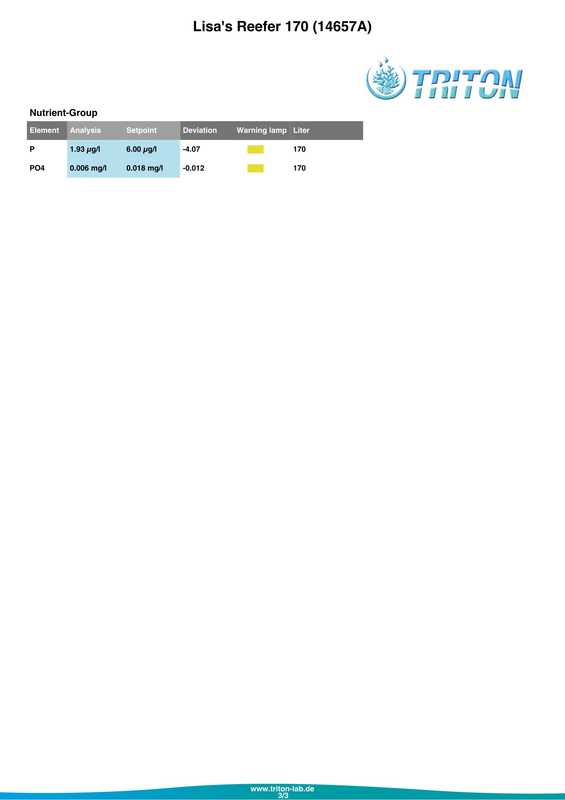
In my quest to discover why some of my corals were looking pale (in one case very pale indeed) with poor or non existent polyp extension I decided to send a sample of tank water off to Triton for an ICP-OES (which stands for Inductively Coupled Plasma-Atomic Emission Spectroscopy) analysis. I must admit that I had actually intended to do this anyway when the tank reached the 6 month mark just because I was curious as to what it would tell me about my water quality. At the same time as sending off the sample I did a whole round of testing myself. The intention being that I could compare both results and see if my test kits (not to mention my testing technique) were up to scratch. Here are the results from my testing performed on the 18th April 2017. Specific gravity: 1.026 (51.7mS according to my conductivity probe) Alkalinity: 7.0dKH (Salifert) Calcium: 410ppm (Salifert) Magnesium: 1272ppm (Salifert) Potassium: 390ppm (Salifert) Strontium: 0-3 (Salifert) Nitrate: undetecable (Salifert) Phosphate: undetectable (Hanna phosphate) Iodide: <0.01 (Salifert) Iodate: <0.03 (Salifert) ...and here are the ICO-OES results. So what do the above results tell me? Well, the obvious issue that immediately jumps out is the elevated tin (Sn) level. Eeek! Where on earth did that come from? I have read that a possible source for tin could be the glass (as molten tin is used to "float" the glass on during it's manufacture). Other than that, something could be rusty and contaminating the system but I'm at a loss as to what it could be, all the equipment was bought new and there's nothing obviously rusting as far as I can see.
Apart from tin, I don't think the rest of the results look too bad really, pretty much as I expected. My tests are not that far off which is good to know (with the possible exception of Strontium which is low with the Salifert test kit but actually at an acceptable level according to Triton). My phosphate level is surprisingly low despite the increased fish load and extra feedings. Triton recommends feeding more and/or reducing the amount of phosphate remover used. As I don't use any remover that's going to be difficult to do but I do intend to feed more to my corals. Results comparison: Me Triton Ca: 410 434 Mg: 1272 1258 K: 390 386 Sr: 0-3 7.06 Iodide/Iodate: <0.01/0.03 0 PO4: undetectable 0.006 So what now? Well I'm going to continue performing water changes as normal, maybe change out a little more in the next a few weeks. Triton recommends 6 x 15%, but they are probably assuming that I don't perform water changes at all which is certainly not the case. I popped a polyfilter into the sump for good measure mainly because I had one to hand (from years ago, I hope they don't 'go off'?) and surely it can't hurt. I also intend to start dosing iodine again. Just before sending off the water sample to Triton as reported in a previous post, I also reduced the intensity of my lighting. It's been 14 days now and I do think that there has been an improvement in the corals, the colours look a tiny bit better and the polyps are starting to extend again. Perhaps the lighting was the main issue? I am hopeful that things will continue to improve. Now I just need to get rid of all that tin.... Photos to follow on Monday with the full May update. For a while now some of the corals have been losing colour. The worst affected being Acropora #3 (A. formosa?), the one that has been recovering from blistered skin. It's reached a point where some form of action needed to be taken. I had hoped that by introducing some more fish the nutrient levels would increase and the corals would look a bit happier, but even though I'm adding a lot more food to the tank the nitrate and phosphate levels still remain oddly undetectable. I performed as many tests as I could on Friday (with the exception of Iodine) and the results were as follows.
Specific gravity: 1.026 Ammonia: 0 Nitrite: 0 Nitrate: 0 Phosphate: 0 Alkalinity: 6.8dKH Calcium: 410ppm Magnesium: 1260ppm Potassium: 380ppm Strontium: 0-3ppm The levels are a touch lower than I'd really like them to be especially alkalinity and Strontium but the Tropic Marin Pro Reef salt mixes up to give an alkalinity level of 6.7dkH and 0-3ppm respectively so if I want those levels to increase I'm going to have to supplement them in my mixing bucket before doing a water change. I intend to add Reef-Roids to feed the corals more frequently as the filtration system I have in place seems able to cope with the extra loading. Along side that I also decided to alter my lighting schedule. I looked at some of the other LX7 lighting profiles posted on the GHL website and their levels were set lower than mine. Had I been unintentionally frying my corals with too much light I wonder? So now I have reduced the levels and the difference is quite noticeable to my eye, the tank looks a lot darker and quite a lot more bluer! To be honest I really don't like the look but if the corals respond positively then that's what counts. I expect I'll get used to it eventually. |
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|


















 RSS Feed
RSS Feed
