|
So one Sunday in May I discovered this: Here's the story. Just after 4am on Sunday morning I woke to the sound of house alarms ringing in the neighbourhood. So annoying! Then it registered that the reason house alarms sometimes trigger is usually related to a power cut, so I groggily reached for my phone to check on the tank. The first thing I noticed was that I'd received a disconnect notification from the return pump which was confirmation that there was a power issue. I tried checking on the tank via the webcam but it wouldn't connect so I tried the bedside lamp. That switched on so clearly the power was restored, I rolled over and went back to sleep. When I got up in the morning the house alarm display was showing an error code but I didn't care about that because immediately after I discovered the water level in the DT was abnormally low. The return pump was not working!! Whatever kind of glitch in the power/internet had knocked out the controller for the return pump and it had not restarted again, this had never happened before. I unplugged it and plugged it back in and water immediately started flowing again. However as soon as water began recirculating the temperature monitors started alarming. I watched as the temperature dropped to 23.5. OK, hopefully that wasn't too bad but 15 minutes later I noticed the tank was starting to look a bit cloudy, uh oh. I checked the refugium and found a significant gathering of mini brittlestars. The temperature dip had triggered them to spawn on mass. I have never seen so many little waving legs in my life, they were everywhere. Up in the DT brittlestars appeared out of every nook and cranny, climbing up literally every coral to spawn. I knew I had a fair few in the tank but I had no idea there were that many. The water got cloudier and cloudier so I started prepping for a water change. Fortunately nothing has been too badly affected by that event but it's not something I'm eager to repeat. I do have multiple temperature probes in case of failure but they are all positioned in the sump. I moved one of the sensors up to the DT so if it happens again I will be alerted but having said that the audible alarm is so puny I doubt I would hear it at night. I should probably invest in another temperature probe for the profilux because when that sounds I most definitely hear it and it scares the life out of me.
0 Comments
It's been almost 8 months since I added the Christmas tree worm rock so time for a proper update. I think things are going pretty well but I don't have a 100% success rate. A few of the worms have vanished (a total of 7 since introduction) for reasons unknown to me. Probably food related but I don't know for sure. The remaining worms (approx 33 of them) seem to be fine, in fact some are bigger and have extended their calcareous tubes quite considerably. Hopefully this is a good sign. I still have all 4 Paguritta crabs (tentatively identified as Paguritta morgani) and the tiny barnacles, spionid worms etc. Sadly I think I have lost the tunicate recently, it was being overgrown by the Cyphastrea and repeatedly covered by sand thanks to a Tigertail cucumber who has taken up residence underneath the rock. I regret not acting sooner to deal with the cucumber/sand situation. The Cyphastrea is growing really well, it has completely covered all the free space on the rock except for one small area which I always found strange. Then, when recording this update, I finally discovered the reason why, there are two teeny tiny gall crabs living in that location. It's only taken me 8 months to discover them, lol (they measure just 2mm wide so are easily missed). As luck would have it one of them shed that very night leaving the empty exoskeleton right by the entrance to his hole so my identification was confirmed. I find them pretty cool but then I find most things reef related fascinating. Since they don't appear to be doing too much damage so I'm going to leave them be. Another short clip of the Paguritta crabs because they are just so cool! Also.... what's that? There's something moving in that hole! My surprising discovery of the gall crab(s?). The video is not the best quality (a bit of an understatement there) but the tiny gall crab can be seen in the area of rock not covered by coral (right of centre). I think there is a second one in another hole above and to the right of the first one. A few macro photos of the gall crab shed exoskeleton that only the most dedicated of reefing nerds are probably interested in, lol. The details are quite hard to make out. Underside: Topside, the carapace looks reddish but that's just algae growth, the new skeleton is whitish is colouration. Side view: View of the crab in her hole the morning after shedding.
I have tentatively identified the hitchhiking large vermatid snail as Dendropoma sp. I recorded this video of the large vermatid snail feeding at the end of December 2021, I had stirred up the sand and noticed it hauling in its mucus web. Generally I'm not keen on vermatid snails (I have an army of the smaller species already and they annoy me intensely) but I find this one kind of cool. It's unlikely to reproduce since (as far as I am aware) I have only one. Unfortunately, a week ago, when feeding the fish their breakfast I noticed a mass of purple coloured mucus like substance covering the bottom right corner of the Christmas tree worm rock. My first thought was I'd lost another one of the Christmas worms but then I realised the mass was centred around the vermatid snail tube. I siphoned away the mucus to discover a dead area of Cyphastrea and no sign of the vermatid snail. Later on the snail showed signs of life, with the operculum again visible at the top of the tube so I wasn't sure what to think. Had another coral released mucus that landed on the Cyphastrea causing the snail to retreat deep down into its tube and suffocated the coral? The next day I discovered one of the Nassarius snails sat right on top of the vermatid snail tube, with another two prowling close by. Clean up crew in action! The vermatid snail was definitely dead then.
I do feel a little sad to lose that snail. Did it starve or was it old, did the mucus kill it or was the mucus a result of it dying? Looking at the video above the snail appeared to be happy enough, eating and producing waste. I assumed it would do just fine without any particular intervention from me, sadly not. Luckily I haven't lost any more Cyphastrea, the immediate area looked a bit unhappy for a few days but seems to have bounced back now. Prior to this the coral was spreading quite nicely so hopefully the dead area will be recovered soon. I haven't lost any of the Christmas tree worms even those that were right next to the vermatid snail and were covered by mucus, I am thankful for that. This is how the area looks a week after the mucus event. Ever since I picked up a Walking Dendro I have wanted to capture a video of the peanut worm hauling the coral around the sand. I have tried on a number of different occasions to record a Timelapse video but the coral never moves, even just a bit. Then, in June last year, I happened to notice the coral was propped up on top of a partially buried Cerith snail with the peanut worm visible below. Yes, I thought, finally a chance to capture some worm action. I hastily set up my phone and clicked record but unfortunately when I checked the footage some 30 minutes later I discovered that the focus was slightly off. I can't tell you how annoyed I was about! Since the video was not the best I never bothered to upload it but here we are 7 months later and I haven't recorded anything better, so what the heck, I'm going to share it anyway. At the beginning of the video the worm can be seen poking out bottom left of the coral and snaking to the right, its head appears just under the sand to the right of the coral. It's going to be hard to make out if you watch on a phone but on a bigger computer screen you can see the star shaped mouth moving right next to the glass. I've fast forwarded through the parts where nothing much happens to make it less boring. Still this video is for die hard peanut worm lovers, you have been warned. Yesterday was pretty eventful, my pair of pink streaked wrasse spawned for the first time. I guess that means that I was fortunate enough to choose a male and female after all. The female had been looking fat all week but yesterday she looked like she'd swallowed a marble, so round was her tummy. The pair seemed to be displaying/interacting a lot and this intensified a couple of hours before lights out so I sat down to watch them. The male would display to the female by swimming on his side next to her and his colouration changed too (I'm not exactly sure to what because the lighting was really, really blue at that time plus I was sitting too far away so not to spook them). The rear of her belly next to the vent went very pale/white. The romancing/courtship went on for about an hour, they would come together, rise to the top of the tank and swim back down again. Finally, about 50 minutes before the lights go out, they did the deed. I downloaded the footage from my webcam but the quality is bad, I mean really, really bad but I'm still pretty excited to have captured it anyway. Maybe when/if it happens again I can try to record it on my phone but the lights will still be blue so it might not be that much better. I have cropped the footage a bit and tried colour correcting some of the blue (also the glass needs a wipe). It's a blink and you'll miss it kind of moment, so if that happens check out time stamp 1:20 (top left of the screen). One of the benefits of the yellow wrasse no longer being present is that whenever the fish are fed and the flow is switched off out pop this pair. I don't know what Flash has been eating recently but his belly is really quite round, lol. An impromptu video of the NPS corals last night after lights out. In hindsight I probably should have wiped the glass and used a tripod. Also apologies for the reflections, the corals look good at least. I was playing around with iMovie in July 2020. Firstly here’s a quick video showing a few random clips of ‘stuff’ in the tank. Milo sends his apologies, he’s regrowing his claw so there’s no clapping today. And secondly there's one fish that doesn't feature very much in my updates because it hides away for most of the time. I don't have many decent photos of him (or her?) as a consequence. I do get to see him almost every day at feeding time and if I don't then I can always rely on hearing his pistol shrimp buddy at some point during the day. The pair are most active after lights out when the rest of the fish are bedded down for the night. This fish may not be out all the time but I don't care because when I get to see him it's all the more special. I really love this fish! Anyway here's a crappy video taken with my phone a couple of nights ago, apologies for the reflections on the glass. It may have been Good Friday (10th April 2020) when this happened but for me it was a BAD Friday. I decided that the time had come for me to move over Clive the Crocea clam from the Reefer, I knew it was going to be stressful for me and undoubtedly for Clive too but I underestimated just how stressful it was going to be for all concerned. Clive has been struggling recently due to being crowded out by corals on all sides and from above too. I had already removed the shading from above (the gorgonian) but the zoanthids growing up his shell were becoming a real issue (again) and I'd put if off for way longer than I should have. I knew that I wanted him off that rock completely and I felt the only way to do that was literally breaking the rock apart. I didn't want to mess around with trying to cut the byssal threads or anything like that. So the whole rock had to come out. Fortunately on set up I'd drilled holes in the rocks and used an acrylic rod to stack them together so only the top rock needed to be removed. Even so that meant pulling out a large expanse of Superman Montipora, the Seriatopora and a whole load of Utter Chaos zoanthids too. Here's the rock in a 25l bucket, Clive can just about be seen in the bottom left hand of the photo. Once I'd pulled off a handful of zoanthids obscuring the lower portion of Clive's shell we got to work on the rock with a hammer and chisel. It was brutal to be honest, the Monti was smashed to pieces and branches of Seri were snapped off right, left and centre. Eventually we ended up with a tiny bit of rock still attached to Clive and his foot appeared to be intact, phew! Then I got to work with a scalpel, scraping off each and every zoa. This is how he looked before his haircut: and here he is afterwards, mission accomplished. He even started to extend his mantle in appreciation. After acclimation I placed Clive in his new home and swiftly opened a bottle of wine to celebrate a job well done except that when I went to check on Clive later I discovered a cloudy tank and found him spawning. My first thought was "Wow how cool, I must record this!" but then as the tank got cloudier and cloudier I began to worry if the filtration of a 2 month old tank could cope with such a large and sudden influx of protein. I googled clam spawning and realised I should probably take action right away. Luckily I already had 20l of saltwater made up already so with the help of my family we began siphoning out the eggs every time they were released. I also set the RO unit to start collecting more water in case extra was needed. Finally Clive was spent and we were too. I tested the ammonia levels 2 hours after the spawning and again after another 2 hours but I couldn't detect elevated levels, thank goodness. I learned something new that day, that clams release sperm AND eggs. The sperm is released first till exhausted and then the eggs are released. Maybe Clive should actually be called Harry/Harriet or something. Clearly this was a stress induced spawning event, sorry Clive. I spent a rather sleepless night in bed worrying if I would get up and find a dead tank in the morning but luckily everything seemed fine. All the fish were present and correct and Clive had his mantle fully extended. He has lost colour in one area due to shading by the zoas but hopefully he can recover that now he's in full light again.
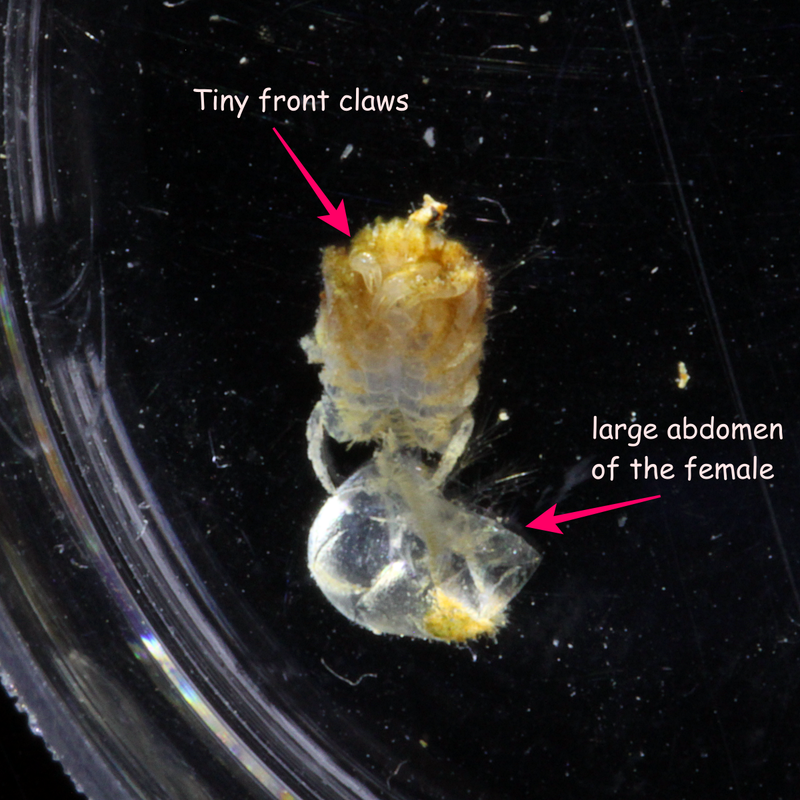
Just a short video of Milo the Venus anemone shrimp (Anclyomenes venustus) hanging out in his Heliofungia home. These shrimp also go by the name of clapping shrimp because when a threat approaches they wave their front claws around frantically back and forth in an amusing clapping motion. |
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|








 RSS Feed
RSS Feed
