|
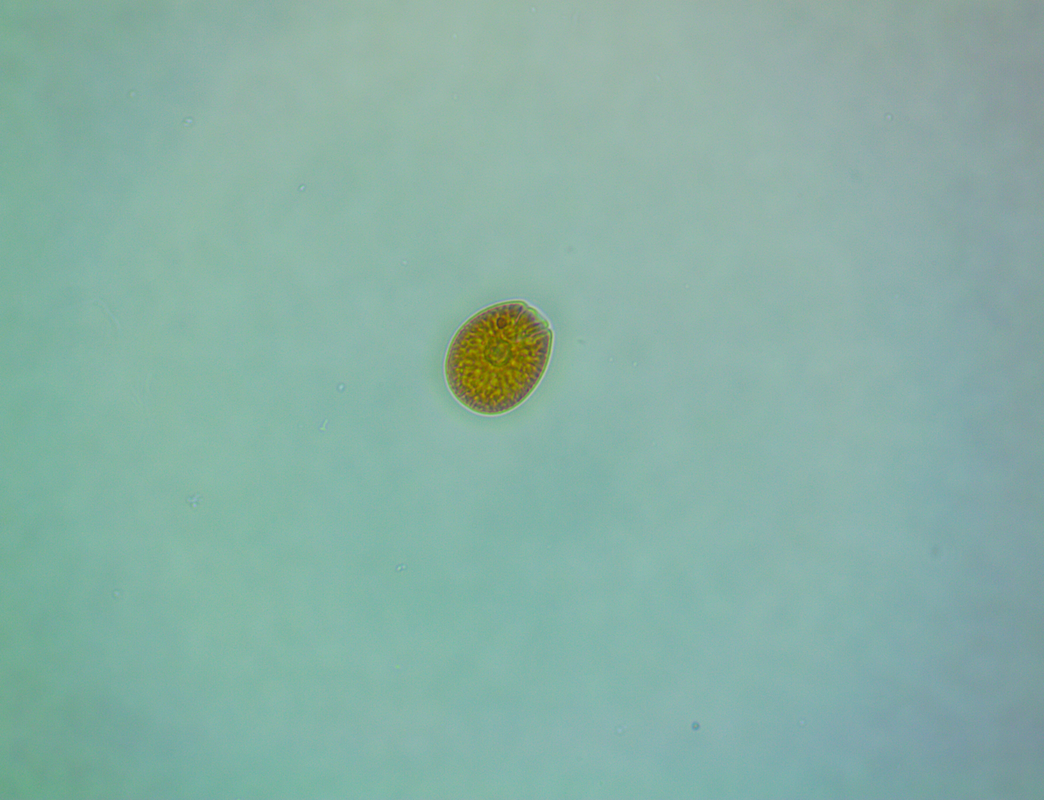
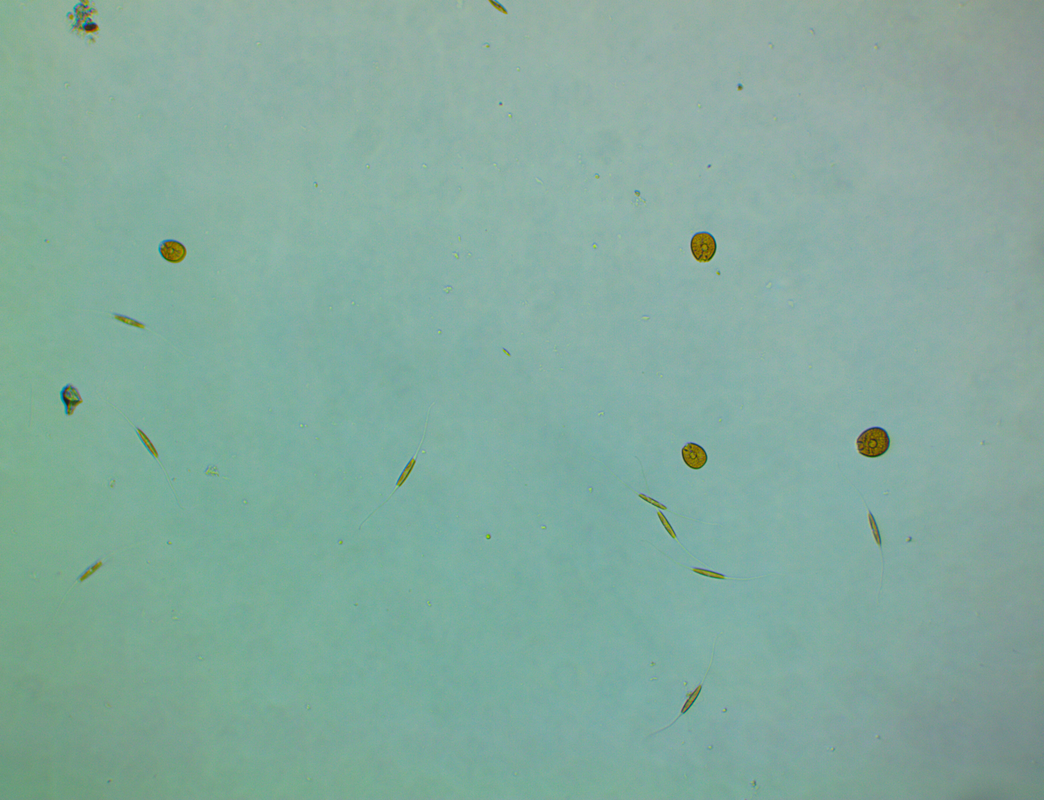
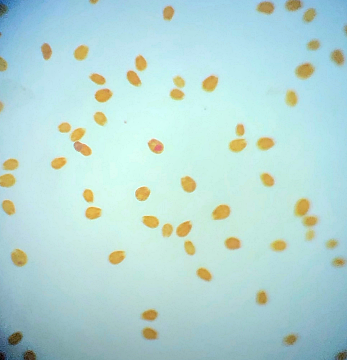
Where to begin? In my last update I mentioned the sand had started to look a bit green and generally manky looking (scientific term, lol). At the start of August I noticed a patch of sand at the back of the tank had developed a suspicious brown look to it. It didn't look quite like diatoms and warning bells were ringing so I dusted off the microscope for a closer look. This is how the sand looked at the back... ...and this is what I saw under the microscope: So yeah it was dinoflagellates but not a species I had encountered before, this was large cell Amphidinium. Apparently this species is not toxic to livestock but can be much harder to get rid of. The best way to tackle it is to outcompete it by encouraging the growth of diatoms, to do that you have to raise the silicate level so I ordered myself a bottle of Brightwell's SpongExcel. I have since found that this is not the cheapest approach as it's not very concentrated, Waterglass is much most cost effective but at least it would get me started. Apart from the dinos making the tank look dirty nothing seemed to be suffering or dying so I took my time with the dosing, starting off slowly. It's pretty hard to test silica levels with home kits, Hanna do make one but it's aimed at freshwater and doesn't work well for saltwater so I didn't see the point of buying it. In addition to silica dosing I decided to get a better grip on the nutrient levels. At the beginning of August the nitrate level was around 2.5ppm thanks to dosing NeoNitro but phosphate was still extremely low often registering as 0 on the Hanna ULR. I invested in some NeoPhos and started dosing that too (bonkers really!). A sand sample taken just over two weeks later looked like this... There were still dinos present but now I was also seeing some diatoms mixed in as well. I continued to dose silica but still at a lower amount than is normally recommended. The phosphate level had risen to approximately 0.04ppm on the Hanna ULR by the end of August. By mid September I realised that the orignal brown patches at the back of the tank had faded away. I sampled the sand on the 11th September I couldn't find any Amphidinium under the microscope, this was honestly quite a surprise to me as I'd expected to see some still hanging around. So my sand was clean right? Well no, I had replaced one sort of brown for another. This was the 'new' brown... I stopped dosing silica and by that time the phosphate was up to 0.06ppm or thereabouts so I decided to hold off dosing any more of that too. By October the sand had progressed from brown to green when cyanobacteria decided to join the party too. It's even got the audacity to smile at me under the microscope! At the time of typing this the sand is a mix of green and brown. I'm not so bothered about the diatoms because the CUC love it but the cyanobacteria is nasty. So how do I combat green Cyano? I did think about using Dr Tim's Re-fresh but it states on the bottle that it could be harmful to snails and shrimps. I queried this with the manufacturer but they wouldn't elaborate as to how serious the risk was or even how the livestock is affected, is it toxic to them or do they die because of a knock on effect? I'd also rather not use chemicals such as Chemiclean either. So I have an imbalance between the nitrate and phosphate levels right? They currently stand at around 4.5 ppm for nitrate (Salifert) and 0.06-0.09ppm phosphate (Hanna ULR, the test results vary from day to day). Should I keep raising the nitrate (I am still dosing NeoNitrate every day) or try and reduce phosphate, or do both? I did add a couple of new fish at the beginning of September in the hope of addressing the nitrate issue but there was no noticeable effect. Maybe I need to add a few more?
The struggle continues...
0 Comments
One of the recently added Trochus snails has sprouted a lush growth of Ulva on his shell. From what I've read this algae seems to be doing the rounds at the moment and since I don't keep any big herbivorous fish, or indeed intend to, it could become a headache for me if/when it spreads. Also I've noticed the appearance of a few patches of what I believe to be green cyanobacteria on the rockwork. I'm hoping this doesn't get any worse. Lastly I discovered another tiny Aiptasia in the tank. . It was growing on the tube of my Coco worm, either it came in with the worm or it has settled onto the tube whilst it's been in my tank, I kind of hope it's the former and not the latter. Where there's one there's probably many more waiting to be discovered. Oh joy! Now for some possibly good news, the Coco worm, Protula bispiralis 'seems' to be doing quite well so far. I'm basing this off of the fact that it's extended it's calcareous tube quite a bit over the last month. If it can lay down some new tube then it must be getting enough to eat, right? When I came to treat the aforementioned Aiptasia with Aiptasia-X, I tried to make it go in first by poking it but despite literally brushing the feathery head three or four times with a pipette it refused to retract. I went ahead and treated the Aiptasia anyway and it stayed out during the entire procedure. I was somewhat concerned by this lack of responsiveness but I just watched a hermit crab crawl over the worm today and it retracted quick as a flash so I guess it simply wasn't bothered enough by me. Here's a few crappy zoomed in iPhone pics showing the tube growth. The first shot was taken on the 3rd June and the second was taken this morning, 4th July, just over a month later. Also I made an exciting discovery whilst performing a water change. I was pumping fresh saltwater into the sump when I noticed some unusual ‘blobs’ moving around down there. On closer inspection I discovered they were baby Trochus snails. How cool is that! OK, I know it’s nothing unusual for snails to spawn in reef tanks but this is the first time I have actually had them settle out and grow into proper baby snails in my tank. So far I have counted 4 of the little chaps but I'm sure there will be more hidden away.
Here’s one of the wee chaps cleaning the base of the skimmer. He’d better not make his way into the pump..... I picked up a frag of macro algae from a fellow reefer. In addition to mopping up a small amount of nutrients I'm hoping it will provide a perfect breeding ground for pods, well that's the plan anyway. The algae is Haliptilon sp. and apparently it can be quite invasive so I opted to keep it away from the rock work. I initially had it sitting on the sand in the right corner of the tank but the snails like to sleep there and have barged it out of the way a number of times. It was also near to one of the entrances of the pistol shrimp burrow and one morning I found it had been almost completely covered by sand. Fortunately it appears quite tough but not wanting to take any chances, I have moved it to the middle rear of the tank now. The hermit crabs have investigated it quite thoroughly and so far it has survived the attention mostly intact but it really needs to be attached to a rock to prevent it being knocked over.
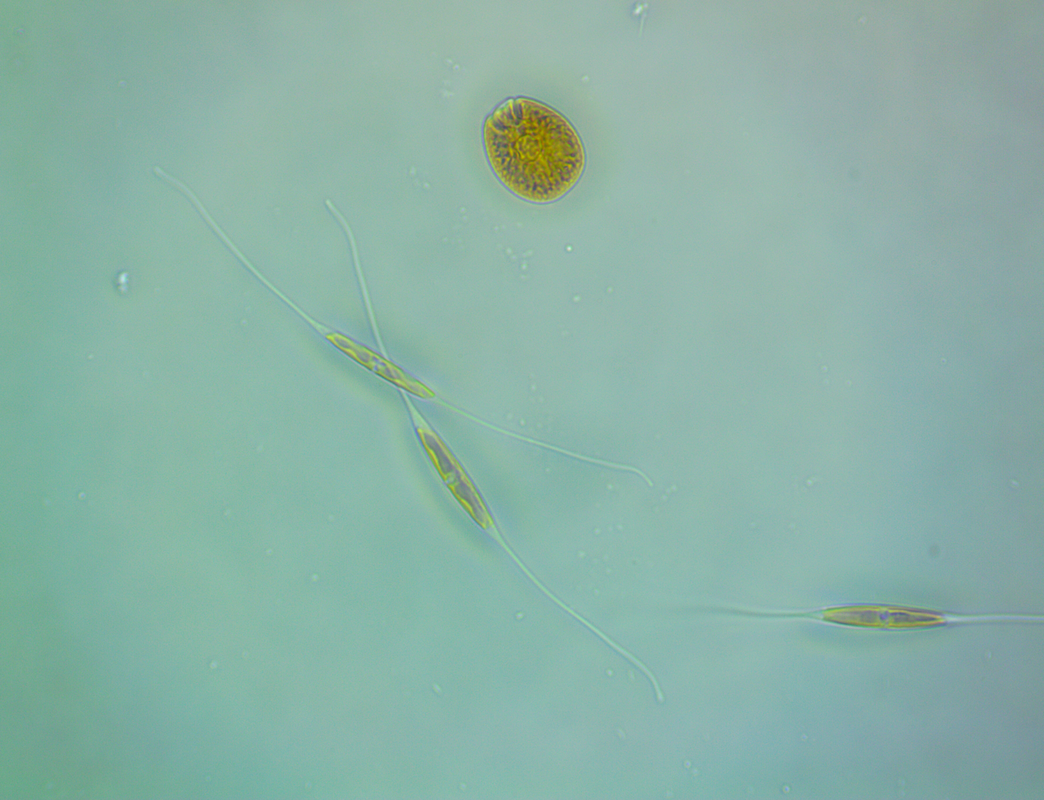
I have lots to update, hmm where to begin? I think I should probably get the bad stuff out of the way first. Forgive me Father for I have sinned. For quite a while I had noticed that I was harvesting less and less macro algae from the refugium, growth had slowed to almost nothing at all despite there being plenty of nitrate and phosphate available. The days of my tank having low nutrients were certainly in the distant past as they had been slowly but surely creeping up. At the last ICP analysis nitrate was sitting at 11.5mg/l and phosphate at 0.13mg/l. I'd also noted that the mini brittle stars that used to thrive in amongst the algae had dwindled from hundreds to zero (there are still lots in the DT). I'd come to the conclusion that Charlize the hitchhiker crab had been supplementing her diet with some brittle star meat. Anyway hair algae had taken a firm hold in the upper half of the refugium and was choking out the growth of macro algae below. This meant that there was even less algae available to the crustacean residents (Charlize and also Bruce the Emerald crab) at the bottom of the refugium. Then at the beginning of December, I discovered what was left of Bruce, had he also been eaten by Charlize? RIP Bruce. So on the 16th December 2019 I decided then to take the refugium out for a good clean, removing the old mud substrate and replacing it with some new, a long overdue task I must admit. I carefully salvaged as much of the macro algae as I could (a mix of Caulerpa racemosa and Chaetomorpha) and placed it, along with the naughty Charlize, into a bucket (with tank water obviously). I then disconnected the refugium and set about cleaning it. Once everything was clean(ish) I added a new layer of mud, refitted the refugium and carefully filled with water. After a while I reintroduced the macro algae and crab. So far so good, however things were about to take an unfortunate downward turn. It seems that by cleaning the refugium and/or replacing the mud I had altered the water chemistry and not in a good way either. The redox value dropped to 250mV and stayed there. Under normal circumstances the redox probe reads somewhere between 350mV to 450mV and to be honest I don't pay that much attention to it. The numbers bounce around depending on whether I've just fed the fish or done a water change. A value of 250mV however was definitely not normal. I wondered if the probe was reading accurately so I cleaned and recalibrated it but still the readings remained low. A few days after cleaning I noticed some suspicious looking brown algae starting to appear in the refugium and I just knew this wasn't going to be good. Sure enough after about a week or so I started to see signs of it appear in the DT too. It began by coating the gorgonians, the Plexaurella was particularly affected and closed up. Ten days after cleaning I discovered Charlize the hitchhiking crab dead in the refugium. Nooo! I couldn't believe it and felt so terrible guilty. Why did she die? My immediate thought was that the brown algae may have been the cause as some species of dinoflagellates are know to be toxic. I've have not been unfortunate enough to have to deal with this type of algae before but I've certainly read about it a lot. It looked just like typical dinoflagellates, brown and snotty with trapped air bubbles but just to be sure I took a sample and dusted off the microscope. My suspicions sadly proved correct. The tiny oval protozoans were swimming in a circular motion around an anchor point like a tetherball which is typical of Ostreopsis sp., this is indeed toxic to snails and other herbivorous creatures. I've read many horror stories regarding dinoflagellates in reef tanks, so to say I was feeling depressed was a bit of an understatement, I had visions of all my corals covered with brown snot and the sand littered with shells of dead snails.
According to my research there is no easy way to rid a tank of dinoflagellates and I certainly wasn't keen on the idea of performing a 5-7 day black out. I decided not to panic and continue tank maintenance as normal. I continued with the weekly water changes (yes I know these were not advised) and siphon out as much of the 'snot' as possible in an effort to give the gorgonians a tiny bit of relief. I don't know if it helped them but it certainly made me feel better. In the ensuing days the dinos spread to the tips of the Seriatopora hystrix but surprisingly nothing else seemed affected. Every morning I would count the number of snails to make sure they were all still present and correct. The redox level remained very low so on the 14th January I sent off a sample of water for ICP analysis just to check if the mud was leeching out something nasty but as you can see from the link below the results looked OK. Nitrates and phosphates were lower than the previous test but not that low. lab.atiaquaristik.com/share/6bba53665864d463b982 Since I'd effectively removed most of the beneficial bacteria and critters from the refugium (except for amphipods and mysid shrimps) I decided it might be a good idea to add some diversity back in the form of some live rock rubble. This proved easier said than done as nobody seems to stock actual live rock anymore, it's all artificial or dead rock and bottled bacteria these days. I tried ordering some in from a local shop but when I went to collect it, it was just a bag of dry rock, sigh! In the end I located an online shop that was out of stock but expecting a fresh delivery of Australian live rock in the next few weeks or so. I decided to preorder a small amount and crossed my fingers that it wouldn't take too long. In the meantime the redox gradually started to creep back up again and by the end of January it had hit 350mV once more, the gorgonians started to pick up again. The first to bounce back was the Muricea followed by the Pinnigorgia and finally after over a month of looking completely dead the Plexaurella shed a layer of algae/mucus and the polyps came back out again, sadly a couple of branches had stripped but at least some of it was still alive. I also only had to frag off one of the Seriatopora branch tips and the rest bounced back in no time at all. As of today the dinoflagellates have almost completely disappeared from the DT, if you check the Pinnigorgia very closely there is still some evidence of some thin brown strings in one area of low flow but there's not much left at all. I am hoping in another month or so it will have gone altogether, I have my fingers crossed. Oh and just as things were starting to look up I received acknowledgement that my small order of live rock was available and ready for delivery (20th February 2019). That story will have to wait until another post, oh the stress is never ending.... A few weeks ago I decided to take action against the hair algae growing on the skeletons of my recently deceased Acros. I armed myself with a toothbrush and tried scrubbing the affected areas, at the same time I performed a water change enabling me to siphon out the algae as soon it was liberated. It didn't take long for me to realise that this just wasn't going to work very well. It was almost impossible to reach some of the areas without running the risk of damaging the surrounding corals and even in the areas I could reach I was unable clear it all off. Worst of all during the process, thanks to my clumsiness, I snapped off the branch tips of several corals. So no more scrubbing for me!
Instead I decided to embrace the hair algae rather than hate it. For years I've wanted to try keeping a Rainford's goby (aka Court Jester goby, Koumansetta rainfordi) but never had the courage to try one because of their reputed difficulty in accepting prepared foods plus need for filamentous algae in their diet. As luck would have it I came across one of these little fish during a visit to a local fish shop, I observed it for a while whilst running through my Rainford's goby purchasing check list. 1. A healthy looking specimen (sleek looking without a concave tummy). ✓ 2. Eating prepared foods (I watched it tuck into frozen mysis). ✓ 3. Mature aquarium to introduce it to (the tank is 22+ months old now). ✓ 4. Filamentous algae present on which it can browse (yes indeed!). ✓ 5. No super aggressive tank mates such as dottybacks or hawkfishes to harass it. ✓ My biggest concern, apart from the feeding issue, was the presence of my Yellow and Pintail wrasse, they are much bigger fish and I had no idea how they would react to the introduction of a small and delicate goby. I was fairly confident that the other nano gobies and wrasse would have no issue. After much umming and aahing I decided to take the risk and bring the little fish home with me. Amazingly, once introduced the new goby was completely ignored by all the resident fish, I was certainly not expecting that! Despite the lack of aggression the goby was far from relaxed which I suppose is normal for any new fish and the Yellow/Pintail wrasses are undoubtedly quite scary to a timid little goby. He didn't hide but kept low down on the sand in the front left-hand corner of the tank and actually looked to be struggling with the brisk flow so I turned down the powerheads to give him some relief. That night he created a cosy little depression in the sand underneath the Utter Chaos zoanthids to sleep in. By the way I have no idea if this fish is male or female, I decided to go with male (a tough little guy with any luck) and named him Jessie. Over the next couple of days Jessie slowly settled in and started exploring the tank. I can confirm that he does peck at/sift the sand and eat hair algae, result! I can't see him being voracious enough to eradicate the hair algae or even keep it in check for that matter but that doesn't bother me right now, I'm just thrilled that he's eating. He was still nervous of the bigger fish and would dart out of their way whenever they approached and when it came to frozen food he would look interested but was not confident enough to grab a bite. Fish have to be quick off the block in this tank when it comes to food as the greedy wrasse tend to hoover it up in no time at all. I increased the number of feedings from three to four times per day and on the fifth day post introduction, Jessie felt confident enough to sneak a couple of small pieces of Gamma Mysis. By the eight day he was up for trying to tackle a large piece of PE Mysis but this was a bit beyond his capability, he tried 'chewing' it 4 times but as he spat it out for yet another attempt a wrasse swooped down and stole it, lol! To ensure that he is getting his fair share I have introduced him to the magical food dispenser (me and a pipette). I like to spot feed most of my livestock (fish, corals, shrimp and crabs) and naturally the fish know that the pipette means food so it takes a bit of time and patience to get the food to the intended target. The Yellow and Pintail wrasse try to steal as much of it as possible, I swear given the chance those fish would keep eating until they popped! I have to wait until these fish 'appear' to lose interest before quickly releasing a piece of food in front of the intended recipient. Fortunately, Jessie is a quick learner and took to the pipette trick remarkably fast, he's even started pecking at the tip in his impatience for the food to appear. It's early days for this fish but so far I think it's looking promising. I will certainly keeping a very close eye on the state of his tummy to make sure it stays full looking and not sucked in. I first discovered Charlize the hitchhiking crab sitting at the base of the Seriatopora in February 2017, she was such a tiny little thing back then no bigger than the nail on my little finger. For a couple of months I let her do her thing before deciding to move her after she knocked a frag off the rockwork. She was surprisingly easy to catch and rather than dispose of her, something I never like to do with any living creature, I placed her into the most desirable of crab residences, the refugium! Since then she has positively thrived and has grown incredibly. She's a beefy crab now measuring a couple of inches across, quite a bit bigger than the Emerald crab she now shares her home with. When I added Bruce (the Emerald crab) I did worry that there might have been issues between the two but so far everything's been good. They tend to hang out at opposite ends of the tank although I occasionally find them relatively close together as in the second photo below. I can't help but wonder how much growing Charlize still has left to do, I may need a bigger refugium if she keeps expanding, gulp!
It occurred to me after my last update regarding the continuing red bug issue that there was one pest I hadn't actually seen in the tank for a while and that was pyramid snails. For months and months I religiously siphoned out as many of the tiny parasitic snails that I could find, I literally removed hundreds of them without any obvious dent in their population. Naturally this got old very fast and as the months went by and my hardworking clean-up crew (Trochus & Turbo snails etc) and clam looked fine I became less vigilant. I would still remove any that I saw attached to the snails but I no longer actively seeked them out. Today though I've searched the tank most thoroughly with a magnifying glass and cannot find any evidence of a single pyramid snail. That's not to say that they aren't still present in the tank but considering how many there were at one point I take it to be a positive sign. Perhaps one of the fish has finally found a taste for them, the most obvious candidate being the Yellow wrasse but I have never seen her (now him) show any interest in eating them even when faced with one crawling up the glass in front of her face. So that's the step forward, now for the backward step. Since the demise of some of my Acropora I have been faced with the issue of what to do with their dead skeletons. I fragged off as much as I could but that still left a goodly amount of encrusted base on the rocks. Sadly these have now become a magnet for hair algae. Normally the snails would have made short work of this algae before it had chance to establish but they find it quite hard to navigate their way round the rockwork these days due to the fact that there are lots of other corals in the way, in fact I hardly ever see the Trochus/Turbo/Ceriths on the rocks at all now, they just spend all their time cruising round the glass. Since this algae is growing ever longer by the day and starting to spread I need to formulate a battle plan asap. That's it for now I'll sign off with an updated video for your viewing pleasure. Not much new to report really, everything seems to be ticking along and there have been no new additions. I harvested a load of macro algae from the refugium as the upper half was a completely solid mass. I think I might have been a bit over zealous with my pruning as the algae growth seems to have stalled somewhat since then. A knock on effect being that the nutrient levels within the tank have risen slightly. On the 9th January phosphate tested at around 0.046ppm and nitrate 0.35ppm using the Elos low range kits. When I tested again on the 10th January the phosphate level had crept up to 0.08ppm and nitrate to 1ppm. Hopefully once the algae growth picks up again the levels will stabilise as I don't really want the phosphate level to get any higher. I must get into the habit of harvesting smaller amounts of algae on a more regular basis. I've also been chasing the male pintail wrasse round in the hopes of capturing a decent photo of him displaying to the female but boy, he's really fast! I have multiple shots of just his tail or the rear half of his body, lol. I am very persistent though, thank goodness for digital cameras or I would have probably given up by now. I finally relented and moved the Balanophyllia to the sump, it was on the fast track to coral heaven if I left it in the DT thanks to the Pintail's attentions. It's so much easier to feed down there now, especially during the day as the refugium is reverse lit so it's nice and dark. The good news is whilst the flesh on one side has receded quite badly due to the earlier infection it doesn't appear to be getting any worse and it's certainly happy to start eating again. It's a pale shadow of what it once was, the photo below shows how far the tentacles currently expand (and the receded skeleton). I hope it improves and opens up fully again. The green tentacled corallimorph was looking a little more extended than I usually see it during the day so I whipped out the camera for a quick snap of that too (with flash). It looks like an anemone (and is commonly called a ball anemone) but actually is a part of the mushroom family. It hitchhiked into my tank on a small piece of zoanthid rock and doesn't appear to have spread much in 8 months time, mind you neither have the zoas either, lol. I think it actually looks rather attractive. I'll sign off with another short video. It's not much different than the last one I posted tbh but hopefully still enjoyable to watch. When the lights switch off above the display tank there's still plenty of action to watch down below in the refugium.
My tank is exactly a year old today! Wow, where has the time flown to? I've been a bit lax with the updates recently so there's lots to catch up on.
First lets get the bad news out of the way. I'm afraid that I've lost the lovely Tridacna maxima clam, it simply never thrived in my tank. Before adding it I carefully checked the shell looking for any nasty hitchhikers but I never found any. I must have missed some (or their eggs at least) as I started to notice some pyramid snails feeding on my precious Trochus and Turbo snails. Nooo! I checked the clam regularly at night with a flash light but never found any of the little devils feasting on it. After two and a half months it finally it became clear that the clam was doomed so I decided to remove it before pollution became an issue. I checked it over again when it was out of the tank and still couldn't find any pyramid snails on it. These guys are so small and clearly hide really well! Later on three teeny tiny ones did emerge from inside of the clam, it's hard to imagine that just three could have any impact on an animal so much larger than they but clearly over time they do. Unless of course the maxima was suffering from something else too? At this point there's not much I can do about the pyramid snails, I am removing any that I see every day and I've become quite adept at spotting them now. On average I remove about 10 per day. The wrasse are sadly not eating them, come on guys I could really do with a helping hand here, sheesh earn your keep why don't you!! At least I haven't lost any of the snails yet and my original Tridacna crocea clam seems to be doing fine, it's laying down new shell so I take that to be a good sign for now.
The other blip on my reefing horizon is the Balanophyllia. It just doesn't look as good as it once did and I can't work out why. Am I feeding it too much or not feeding it enough? I had been offering it a piece of food once per day (at night), generally PE mysis, clam, Krill or lancefish, perhaps that wasn't enough to sustain it? So I decided to up the feedings to multiple times per day (anywhere between three up to a maximum of five a day) but this seemed to make no difference at all (in fact the coral looked a little worse) so now I'm trying less food. It's really frustrating because the sun coral is looking fantastic on a single feed per day.
Apart from the above everything else seems to be doing OK. The fish are all good, Rei the Yellow wrasse eats like a horse and is noticeably bigger. The best news is that my Tomiyamichthys nudus gobies have finally paired up with the Red Spotted pistol shrimp so I get to see them all much more now. The male goby still goes MIA every now and again but always resurfaces at some point. The gobies and pistol shrimp do not naturally associate together in the wild but I suspect they have done so in my tank because there are simply no other alternatives.
The corals are getting bigger and some are starting to get close to each other already, war is on the horizon I expect.
The zoanthids are spreading nicely especially the Utter Chaos, these are reproducing at a phenomenal rate and unfortunately over taking some of the original slower growing morphs. Whatever was afflicting the Red Tuxedo zoanthids seems to have subsided and I've not lost any more recently, I hope that's the end of that.
After a bit of a slow start the algae in the refugium has really got going now and the amount of life in there is incredible. It's amphipod, mysis shrimp and brittlestar heaven! Charlie the hitchhiking crab is alive and kicking and still growing. She was such a tiny thing when I first noticed her in the DT hanging out in the Seriatopora, now she's huge in comparison.
Life in the refugium.
Whenever I harvest any algae, I spend the following 30 minutes rescuing brittlestars from amongst the fronds. Well I can't just throw them out can I? It's easy to see how they are reproducing by division.
My first canister of ATI Carbo EX came to the end of its life in September, it lasted just over 3 months which I don't think is too bad. I have decided to continue with the CO2 scrubbing and have replaced it with a fresh cartridge.
Last week to celebrate the fact that the tank was approaching its first birthday I decided that some new additions were required. There was a gap (left by the T. maxima clam) that was just crying out to be filled. OK it didn't really need to be filled but what can I say, any excuse to shop for new corals.
I decided another encrusting Montipora sp. would do nicely and since it was likely to be the last addition (never say never tho) I wanted something special. I decided the Beach Bum (what a name!!) Montipora would contrast nicely with the three that I currently have. Since I was mail ordering from a fellow reefer I found I couldn't just buy the one coral so I ended up with frags of Hawkins Echinata (Acropora echinata) and a Sunrise Goniopora too. My name is Lisa and I'm a coral addict, lol! Here they are on the sand awaiting fixing (squeezing!) in place.
Phew that was a marathon (are you still with me?) before I sign off I'll add a few more updated photos. After all who doesn't love a bit of eye candy?!
|
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|



















































 RSS Feed
RSS Feed
