|
So one Sunday in May I discovered this: Here's the story. Just after 4am on Sunday morning I woke to the sound of house alarms ringing in the neighbourhood. So annoying! Then it registered that the reason house alarms sometimes trigger is usually related to a power cut, so I groggily reached for my phone to check on the tank. The first thing I noticed was that I'd received a disconnect notification from the return pump which was confirmation that there was a power issue. I tried checking on the tank via the webcam but it wouldn't connect so I tried the bedside lamp. That switched on so clearly the power was restored, I rolled over and went back to sleep. When I got up in the morning the house alarm display was showing an error code but I didn't care about that because immediately after I discovered the water level in the DT was abnormally low. The return pump was not working!! Whatever kind of glitch in the power/internet had knocked out the controller for the return pump and it had not restarted again, this had never happened before. I unplugged it and plugged it back in and water immediately started flowing again. However as soon as water began recirculating the temperature monitors started alarming. I watched as the temperature dropped to 23.5. OK, hopefully that wasn't too bad but 15 minutes later I noticed the tank was starting to look a bit cloudy, uh oh. I checked the refugium and found a significant gathering of mini brittlestars. The temperature dip had triggered them to spawn on mass. I have never seen so many little waving legs in my life, they were everywhere. Up in the DT brittlestars appeared out of every nook and cranny, climbing up literally every coral to spawn. I knew I had a fair few in the tank but I had no idea there were that many. The water got cloudier and cloudier so I started prepping for a water change. Fortunately nothing has been too badly affected by that event but it's not something I'm eager to repeat. I do have multiple temperature probes in case of failure but they are all positioned in the sump. I moved one of the sensors up to the DT so if it happens again I will be alerted but having said that the audible alarm is so puny I doubt I would hear it at night. I should probably invest in another temperature probe for the profilux because when that sounds I most definitely hear it and it scares the life out of me.
0 Comments
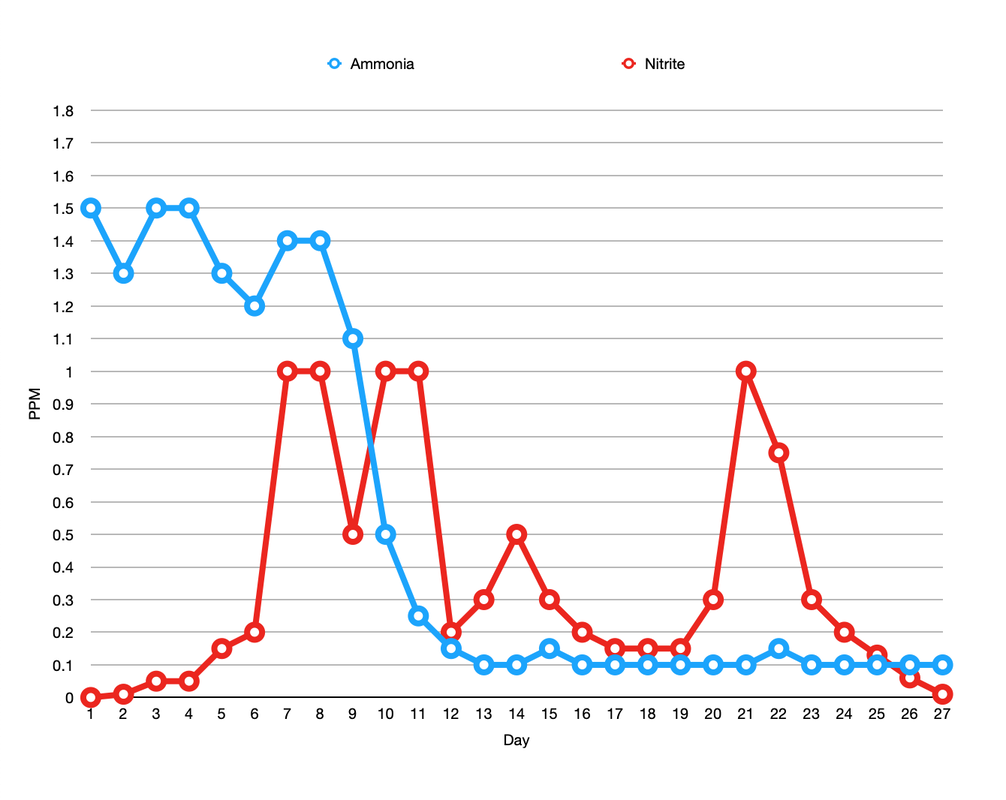
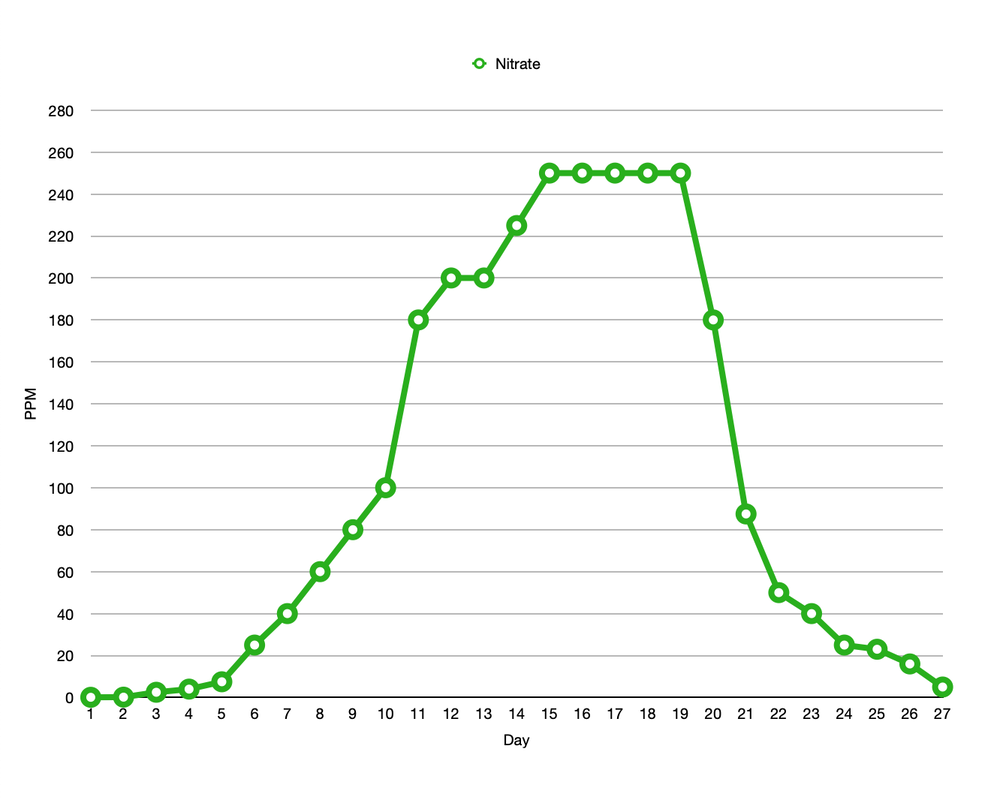
I began the tank cycle on the 2nd February 2020 with a bottle of Dr Tim's One and Only plus the recommended dose of ammonium chloride. Thereafter I tested ammonia, nitrite and nitrate every day for 27 days. Needless to say the levels I recorded in my tank did not look like the neat and tidy graphs I see posted online. I added further doses of ammonium chloride, as per instructions, on days 3 and 6. I also boosted the ammonia again on day 15 and 17 to keep things ticking over. The lights were left off during the cycle but there was some overspill from the Reefer 170. Here's what the ammonia and nitrite levels looked like. And here is nitrate. By day 10 the nitrate level had reached the upper limit of my Salifert test kit so I had to dilute the sample first by 1 in 5 and then later on by 1 in 10. On day 15 the nitrate level had reached a whopping 250ppm!! I envisioned having to perform water changes every week for the next 6 months or so. Things were starting to look a bit grim. The largest water change I can perform at any one time is 30% so on day 19 that's exactly what I did. The following day I recorded the level at 180ppm which is exactly the amount I expected following the water change but what I didn't expect was to see an increase in the nitrite level again. I continued my daily testing and not only did the nitrite levels fall back down again but the nitrate levels kept dropping and dropping till on day 27 they read a rather acceptable 5ppm with no further water changes required. How weird! I have a feeling that the off the scale nitrate levels were probably due to interference by nitrite, either that or I have a super duper nitrate reducing system in my tank. Patent pending. Whilst the tank was cycling my tank cover arrived and I was somewhat disappointed to discover that there is what I consider to be an excessively large gap between top of the weir comb and the mesh lid. What's that all about? Why can't the level of the weir be designed to be more in line with the top of the tank?? I don't have the D-D branded cover but I can't imagine that sits any lower than any of the other alternatives. Anyway there's more than enough room for any of my small to moderately sized fish to squeeze through and since there is no cover on the weir they'll end up stuck down inside it or take a ride down into the sump. Neither alternative is acceptable so I plugged the gap with some clear acrylic sheet. There is still a small gap to the right of the weir comb that I haven't fixed but it's pretty narrow so hopefully my fish won't discover it (famous last words I know). On day 11 I fired up the skimmer, an Ultra-Reef UKS-160 mainly for aeration purposes. This is not a new skimmer but you wouldn't know it, as soon as the pump was fired up it went completely crazy so I left the cup off for the time being. Plenty of air going in there to be sure. 10 days later it showed no signs of calming down and I was getting a bit sick of the salt creep everywhere so I fitted the cup with the drain plug removed so that the water overflowed into the cup and back into the sump again. As you can see protein scum began collecting inside the cup. Skip forward another 13 days and still the skimmer was in overdrive despite the flow restrictor being wide open and the unit sitting in exactly the right level of water. I decide to craft a stand to raise it up a bit, eggcrate, PVC pipe and cable ties later...ta da! That was version 1, since then the pipe legs have been cut down a bit as the skimmer was then sitting a bit too high (typical) but it's started working well now. I have read that skimmer collection is reduced in tanks where roller filters are also employed but it's definitely pulling out some stinky stuff. Talking of the X-filter, I must hold my hand up and admit how I eagerly watched and waited for the motor to turn for the very first time, yes I'm sad like that. It seemed to take an age for the water level to rise up and trigger the level sensor, lol. The excitement has since worn off a bit as I've become used to the motor just doing its thing, it is quite satisfying to see the used dirty brown roll. Better out than in (I hope). On day 27 I considered the tank to be pretty much cycled. Diatoms were beginning to take hold on the sand and rocks so I decided to add some Tisbe copepods. I wanted to encourage the growth of these little critters as much as possible before transferring any of (my pod loving) fish over from the Reefer. For a couple of days I thought the tank must be toxic and I'd killed them all off as I couldn't find a single pod on the glass but then they bit by bit they slowly started to appear. This made me one extremely happy reefer (I'm easily pleased clearly). On the same day as adding the copepods I fired up the refugium. I'm a big fan of refugia not just as an area for the growth of macro algae but also for the reproduction of beneficial critters (and also as a place to relegate any naughty crabs etc if needed). I wanted to keep the light spill down to a minimum inside the sump itself so I designed something to sit in the cupboard to the left of the sump with the pipework entering and exiting via the existing hole in the cabinet. I didn't want to have to modify the cabinet in any way and I almost, just almost, got away with it. The pipework was a tight fit but worked out perfectly, however the tank height was out by just over a millimetre, arghh! I had to sand down the back wall of the cabinet a touch to get it to fit. Ooops! I decided not to make the tank myself this time as it would be a lot bigger than my previous set up and hence more water to worry about leaking, instead I got Wharf Aquatics to make it for me. I'm running a Kessil H80 for the time being (that one I had lighting the Reefer refugium) but I may switch to something more viewer friendly at a later date, I'm not really a fan of the red/purple lighting to be honest. Here it is looking clean and tidy (needless to say it doesn't look like this now, far from it!). Incidentally even though I did not directly add any copepods to the refugium some have made it down there anyway from the DT and it's now buzzing with pod activity. I have the X-filter bypass controllers fully closed but still the pods have managed to find a way through the filter, past the skimmer and through the refugium pump/pipe and into the refugium, which I find pretty amazing to be honest.
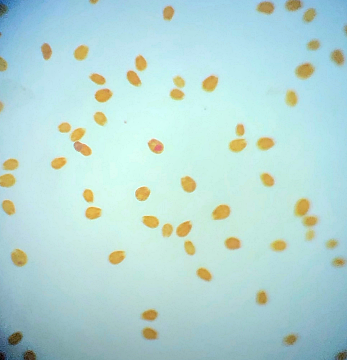
I have lots to update, hmm where to begin? I think I should probably get the bad stuff out of the way first. Forgive me Father for I have sinned. For quite a while I had noticed that I was harvesting less and less macro algae from the refugium, growth had slowed to almost nothing at all despite there being plenty of nitrate and phosphate available. The days of my tank having low nutrients were certainly in the distant past as they had been slowly but surely creeping up. At the last ICP analysis nitrate was sitting at 11.5mg/l and phosphate at 0.13mg/l. I'd also noted that the mini brittle stars that used to thrive in amongst the algae had dwindled from hundreds to zero (there are still lots in the DT). I'd come to the conclusion that Charlize the hitchhiker crab had been supplementing her diet with some brittle star meat. Anyway hair algae had taken a firm hold in the upper half of the refugium and was choking out the growth of macro algae below. This meant that there was even less algae available to the crustacean residents (Charlize and also Bruce the Emerald crab) at the bottom of the refugium. Then at the beginning of December, I discovered what was left of Bruce, had he also been eaten by Charlize? RIP Bruce. So on the 16th December 2019 I decided then to take the refugium out for a good clean, removing the old mud substrate and replacing it with some new, a long overdue task I must admit. I carefully salvaged as much of the macro algae as I could (a mix of Caulerpa racemosa and Chaetomorpha) and placed it, along with the naughty Charlize, into a bucket (with tank water obviously). I then disconnected the refugium and set about cleaning it. Once everything was clean(ish) I added a new layer of mud, refitted the refugium and carefully filled with water. After a while I reintroduced the macro algae and crab. So far so good, however things were about to take an unfortunate downward turn. It seems that by cleaning the refugium and/or replacing the mud I had altered the water chemistry and not in a good way either. The redox value dropped to 250mV and stayed there. Under normal circumstances the redox probe reads somewhere between 350mV to 450mV and to be honest I don't pay that much attention to it. The numbers bounce around depending on whether I've just fed the fish or done a water change. A value of 250mV however was definitely not normal. I wondered if the probe was reading accurately so I cleaned and recalibrated it but still the readings remained low. A few days after cleaning I noticed some suspicious looking brown algae starting to appear in the refugium and I just knew this wasn't going to be good. Sure enough after about a week or so I started to see signs of it appear in the DT too. It began by coating the gorgonians, the Plexaurella was particularly affected and closed up. Ten days after cleaning I discovered Charlize the hitchhiking crab dead in the refugium. Nooo! I couldn't believe it and felt so terrible guilty. Why did she die? My immediate thought was that the brown algae may have been the cause as some species of dinoflagellates are know to be toxic. I've have not been unfortunate enough to have to deal with this type of algae before but I've certainly read about it a lot. It looked just like typical dinoflagellates, brown and snotty with trapped air bubbles but just to be sure I took a sample and dusted off the microscope. My suspicions sadly proved correct. The tiny oval protozoans were swimming in a circular motion around an anchor point like a tetherball which is typical of Ostreopsis sp., this is indeed toxic to snails and other herbivorous creatures. I've read many horror stories regarding dinoflagellates in reef tanks, so to say I was feeling depressed was a bit of an understatement, I had visions of all my corals covered with brown snot and the sand littered with shells of dead snails.
According to my research there is no easy way to rid a tank of dinoflagellates and I certainly wasn't keen on the idea of performing a 5-7 day black out. I decided not to panic and continue tank maintenance as normal. I continued with the weekly water changes (yes I know these were not advised) and siphon out as much of the 'snot' as possible in an effort to give the gorgonians a tiny bit of relief. I don't know if it helped them but it certainly made me feel better. In the ensuing days the dinos spread to the tips of the Seriatopora hystrix but surprisingly nothing else seemed affected. Every morning I would count the number of snails to make sure they were all still present and correct. The redox level remained very low so on the 14th January I sent off a sample of water for ICP analysis just to check if the mud was leeching out something nasty but as you can see from the link below the results looked OK. Nitrates and phosphates were lower than the previous test but not that low. lab.atiaquaristik.com/share/6bba53665864d463b982 Since I'd effectively removed most of the beneficial bacteria and critters from the refugium (except for amphipods and mysid shrimps) I decided it might be a good idea to add some diversity back in the form of some live rock rubble. This proved easier said than done as nobody seems to stock actual live rock anymore, it's all artificial or dead rock and bottled bacteria these days. I tried ordering some in from a local shop but when I went to collect it, it was just a bag of dry rock, sigh! In the end I located an online shop that was out of stock but expecting a fresh delivery of Australian live rock in the next few weeks or so. I decided to preorder a small amount and crossed my fingers that it wouldn't take too long. In the meantime the redox gradually started to creep back up again and by the end of January it had hit 350mV once more, the gorgonians started to pick up again. The first to bounce back was the Muricea followed by the Pinnigorgia and finally after over a month of looking completely dead the Plexaurella shed a layer of algae/mucus and the polyps came back out again, sadly a couple of branches had stripped but at least some of it was still alive. I also only had to frag off one of the Seriatopora branch tips and the rest bounced back in no time at all. As of today the dinoflagellates have almost completely disappeared from the DT, if you check the Pinnigorgia very closely there is still some evidence of some thin brown strings in one area of low flow but there's not much left at all. I am hoping in another month or so it will have gone altogether, I have my fingers crossed. Oh and just as things were starting to look up I received acknowledgement that my small order of live rock was available and ready for delivery (20th February 2019). That story will have to wait until another post, oh the stress is never ending.... I first discovered Charlize the hitchhiking crab sitting at the base of the Seriatopora in February 2017, she was such a tiny little thing back then no bigger than the nail on my little finger. For a couple of months I let her do her thing before deciding to move her after she knocked a frag off the rockwork. She was surprisingly easy to catch and rather than dispose of her, something I never like to do with any living creature, I placed her into the most desirable of crab residences, the refugium! Since then she has positively thrived and has grown incredibly. She's a beefy crab now measuring a couple of inches across, quite a bit bigger than the Emerald crab she now shares her home with. When I added Bruce (the Emerald crab) I did worry that there might have been issues between the two but so far everything's been good. They tend to hang out at opposite ends of the tank although I occasionally find them relatively close together as in the second photo below. I can't help but wonder how much growing Charlize still has left to do, I may need a bigger refugium if she keeps expanding, gulp!
I decided that the clean-up-crew needed a bit of a boost. Whilst I still have two original Ceriths introduced in November 2016, the pair I added 6 months later are no longer with me. I don't get to see these snails very much as they only come out to play when the lights go out but I find they are hard workers and (in a very, very small way) useful keeping the sand turned. So I introduced 6 more. In addition to which I added 5 more teeny-tiny Blue Leg hermits (complement the two already present) and, after much deliberation, an Emerald crab. I have wanted to keep an Emerald crab for ages but have always wimped out of adding one at the very last minute, this time however I actually went ahead and did the deed. I had hoped that it would deal with an ever growing number of Valonia (bubble algae) that were popping up on one particular piece of the rockwork. On day one the Emerald crab (Bruce, as he is now named) picked at the rockwork (good) and sampled the Superman Montipora and Seriatopora hystrix (not good!). On day two some of the small fan worms that I'd been carefully nurturing disappeared but I'd kinda expected this, what I didn't expect however was to wake up to 3 complete branches of Seriatopora stripped of flesh. Alarm bells were definitely ringing. Day 3, things looked worse for the Seriatopora, losing fans worms is one thing but eating corals is a no-no so a quick herding with a pipette into a glass cup (not too bright this crab) and down into the refugium he went. Now Bruce spends his days in bliss and munching on the veggies like he's supposed to. Sadly he never got the chance to deal with growing number of Valonia in the DT. Here's a quick pic from day 1 before he blotted his copy book: And now here's a quick video of him living it up in the veg paradise that is the refugium, those grapes are oh so tasty (but maybe not so much as coral, rofl). Apologies for the freaky red lighting, it's not great for videos but the algae seems OK to grow under it. Firstly another equipment change to report. I finally decided to change out the Beamswork EVO refugium light for a Kessil H80 Tuna flora. I had been considering replacing it for quite a while now, not because I was unhappy with the light but because the metal stand was rusting in all four corners where it sits on the top of the refugium. I'm not sure if this has caused any metal contamination in the tank yet or not but I wasn't happy with the situation so it had to go. The reason I opted for the Kessil is because it's pretty much the only light that I could find small enough to fit over the refugium, everything else was either too big/long and would have caused a huge light overspill issue or excess heat in the small sump area. To combat at least some of the light spill produced by the H80 I have added a small piece of black acrylic sheet between it and rest of the sump. The red light is something that does take a bit of getting used to but if the macro algae grows well under it then I'm happy, time will tell. Now for a Fauna Marin Skimbreeze update. The FM media lasted two and a half weeks before needing to be replaced, about the same amount of time as the most recent ATI 1l disposable unit. I'm not that keen on the design of this reactor itself as air is preferentially pulled from the side the outlet tube sits (as can be seen in the photo below). So when changing out the reactor some media is still unused. I think I will try rotating the cylinder next time to see if I can reduce the problem. To refill the reactor I used the Spherasorb media, it looks exactly the same as the FM stuff but costs a lot less. As for the livestock well everything is ticking along. Acropora growth is slow to nonexistent thanks to the red bugs but at least they are not dying. The exception to this is the A. hyacinthus which seems fine (with a rather odd growth shape it must be said), I've accidentally fragged it with the algae magnet a couple of times now because it's growing too close to the front glass. The Utter chaos zoanthids are becoming annoying as they keep detaching and floating around the tank ending up in spots where I don't want them in or sucked on to the powerheads. Since I don't have anywhere to place frags (and hate the look of frag racks in the DT) I've chucked the loose ones I've found so far. The sun coral is still releasing planulae and I keep finding baby polyps popping all over the tank. The more I look the more I can spot, they've even settled out on the clam shell. The larvae are fascinating to watch, tiny orange tear shaped blobs that wiggle around the tank until they find somewhere to settle out. It's very cool! As for the Balanophyllia now living down in the sump, I can't decide if it's doing better or worse. There is one thing though, I happened to knock it over whilst trying to feed it and when I set it straight again I noticed it has some tentacles poking out the back, my initial response was Oh ****! Aiptasia!! But on closer inspection it actually looks like a baby Balano bud. I'm not sure if this a good sign or a bad one? Is it failing so badly that it's sending out buds as a last ditch attempt to survive?? I surely hope not. I'll sign off with a few photos. And finally a FTS..
Not much new to report really, everything seems to be ticking along and there have been no new additions. I harvested a load of macro algae from the refugium as the upper half was a completely solid mass. I think I might have been a bit over zealous with my pruning as the algae growth seems to have stalled somewhat since then. A knock on effect being that the nutrient levels within the tank have risen slightly. On the 9th January phosphate tested at around 0.046ppm and nitrate 0.35ppm using the Elos low range kits. When I tested again on the 10th January the phosphate level had crept up to 0.08ppm and nitrate to 1ppm. Hopefully once the algae growth picks up again the levels will stabilise as I don't really want the phosphate level to get any higher. I must get into the habit of harvesting smaller amounts of algae on a more regular basis. I've also been chasing the male pintail wrasse round in the hopes of capturing a decent photo of him displaying to the female but boy, he's really fast! I have multiple shots of just his tail or the rear half of his body, lol. I am very persistent though, thank goodness for digital cameras or I would have probably given up by now. I finally relented and moved the Balanophyllia to the sump, it was on the fast track to coral heaven if I left it in the DT thanks to the Pintail's attentions. It's so much easier to feed down there now, especially during the day as the refugium is reverse lit so it's nice and dark. The good news is whilst the flesh on one side has receded quite badly due to the earlier infection it doesn't appear to be getting any worse and it's certainly happy to start eating again. It's a pale shadow of what it once was, the photo below shows how far the tentacles currently expand (and the receded skeleton). I hope it improves and opens up fully again. The green tentacled corallimorph was looking a little more extended than I usually see it during the day so I whipped out the camera for a quick snap of that too (with flash). It looks like an anemone (and is commonly called a ball anemone) but actually is a part of the mushroom family. It hitchhiked into my tank on a small piece of zoanthid rock and doesn't appear to have spread much in 8 months time, mind you neither have the zoas either, lol. I think it actually looks rather attractive. I'll sign off with another short video. It's not much different than the last one I posted tbh but hopefully still enjoyable to watch. When the lights switch off above the display tank there's still plenty of action to watch down below in the refugium.
My tank is exactly a year old today! Wow, where has the time flown to? I've been a bit lax with the updates recently so there's lots to catch up on.
First lets get the bad news out of the way. I'm afraid that I've lost the lovely Tridacna maxima clam, it simply never thrived in my tank. Before adding it I carefully checked the shell looking for any nasty hitchhikers but I never found any. I must have missed some (or their eggs at least) as I started to notice some pyramid snails feeding on my precious Trochus and Turbo snails. Nooo! I checked the clam regularly at night with a flash light but never found any of the little devils feasting on it. After two and a half months it finally it became clear that the clam was doomed so I decided to remove it before pollution became an issue. I checked it over again when it was out of the tank and still couldn't find any pyramid snails on it. These guys are so small and clearly hide really well! Later on three teeny tiny ones did emerge from inside of the clam, it's hard to imagine that just three could have any impact on an animal so much larger than they but clearly over time they do. Unless of course the maxima was suffering from something else too? At this point there's not much I can do about the pyramid snails, I am removing any that I see every day and I've become quite adept at spotting them now. On average I remove about 10 per day. The wrasse are sadly not eating them, come on guys I could really do with a helping hand here, sheesh earn your keep why don't you!! At least I haven't lost any of the snails yet and my original Tridacna crocea clam seems to be doing fine, it's laying down new shell so I take that to be a good sign for now.
The other blip on my reefing horizon is the Balanophyllia. It just doesn't look as good as it once did and I can't work out why. Am I feeding it too much or not feeding it enough? I had been offering it a piece of food once per day (at night), generally PE mysis, clam, Krill or lancefish, perhaps that wasn't enough to sustain it? So I decided to up the feedings to multiple times per day (anywhere between three up to a maximum of five a day) but this seemed to make no difference at all (in fact the coral looked a little worse) so now I'm trying less food. It's really frustrating because the sun coral is looking fantastic on a single feed per day.
Apart from the above everything else seems to be doing OK. The fish are all good, Rei the Yellow wrasse eats like a horse and is noticeably bigger. The best news is that my Tomiyamichthys nudus gobies have finally paired up with the Red Spotted pistol shrimp so I get to see them all much more now. The male goby still goes MIA every now and again but always resurfaces at some point. The gobies and pistol shrimp do not naturally associate together in the wild but I suspect they have done so in my tank because there are simply no other alternatives.
The corals are getting bigger and some are starting to get close to each other already, war is on the horizon I expect.
The zoanthids are spreading nicely especially the Utter Chaos, these are reproducing at a phenomenal rate and unfortunately over taking some of the original slower growing morphs. Whatever was afflicting the Red Tuxedo zoanthids seems to have subsided and I've not lost any more recently, I hope that's the end of that.
After a bit of a slow start the algae in the refugium has really got going now and the amount of life in there is incredible. It's amphipod, mysis shrimp and brittlestar heaven! Charlie the hitchhiking crab is alive and kicking and still growing. She was such a tiny thing when I first noticed her in the DT hanging out in the Seriatopora, now she's huge in comparison.
Life in the refugium.
Whenever I harvest any algae, I spend the following 30 minutes rescuing brittlestars from amongst the fronds. Well I can't just throw them out can I? It's easy to see how they are reproducing by division.
My first canister of ATI Carbo EX came to the end of its life in September, it lasted just over 3 months which I don't think is too bad. I have decided to continue with the CO2 scrubbing and have replaced it with a fresh cartridge.
Last week to celebrate the fact that the tank was approaching its first birthday I decided that some new additions were required. There was a gap (left by the T. maxima clam) that was just crying out to be filled. OK it didn't really need to be filled but what can I say, any excuse to shop for new corals.
I decided another encrusting Montipora sp. would do nicely and since it was likely to be the last addition (never say never tho) I wanted something special. I decided the Beach Bum (what a name!!) Montipora would contrast nicely with the three that I currently have. Since I was mail ordering from a fellow reefer I found I couldn't just buy the one coral so I ended up with frags of Hawkins Echinata (Acropora echinata) and a Sunrise Goniopora too. My name is Lisa and I'm a coral addict, lol! Here they are on the sand awaiting fixing (squeezing!) in place.
Phew that was a marathon (are you still with me?) before I sign off I'll add a few more updated photos. After all who doesn't love a bit of eye candy?!
Charlie climbed to the top of the refugium to say hello to me this morning. I discovered her sitting on top of the thick mat of algae. Oh my, she's grown a lot! So is clearly finding enough to eat down there. :o)
|
AuthorHi, my name is Lisa and I live in Derby, UK. I am a self-confessed reefaholic! Archives
July 2022
Categories
All
|





















































































 RSS Feed
RSS Feed
